New user? / Forgot your password?
0 Item | 0,00 €
Amisulpride – Reference Materials for Residue and Environmental Analysis
Amisulpride PRECISION Reference Materials for Confident Compliance
Ensure accuracy from method development to routine monitoring with high-purity Amisulpride reference materials from HPC Standards. Our certified neat solids and ready-to-use solutions are manufactured and tested to international quality requirements, enabling robust LCMSMS quantification across water, wastewater, sludge, sediments, biota, clinical matrices, and pharmaceutical QC. Benefit from matrix-matched performance with optional stable isotopelabelled internal standards, documented traceability, and lot-specific CoAs supporting ISOIEC 17025 and ISO 17034 workflows. Custom concentrations and dependable stability data help your lab achieve consistent recoveries, defensible results, and streamlined auditson time, every time.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Certified Reference Material Amisulpride solution | 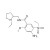 | 687449 | 1X1ML | Please log in. |
ISO 17034 Reference Material | 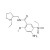 | 691613 | 1X100MG | Please log in. |
High-quality reference materials for Amisulpride to support precise quantification in food, environmental, and pharmaceutical residue analysis. Manufactured and tested to international quality requirements for reliable compliance and monitoring.
Overview
Amisulpride is a substituted benzamide and atypical antipsychotic targeting dopamine D2/D3 receptors. It is widely measured in clinical, environmental, and residue-analysis contexts due to its therapeutic use and potential occurrence in wastewater and surface waters. Accurate quantification is essential for regulatory compliance, exposure assessment, and method validation.
Chemical Identity & Properties
Class: Substituted benzamide; therapeutic group: atypical antipsychotic (dopamine antagonist). The compound is polar with basic functional groups and is typically analyzed as the parent molecule. Physicochemical features (e.g., moderate water solubility depending on salt form, positive ionization under ESI+) enable sensitive LC–MS/MS workflows. Handle as light- and temperature-sensitive according to best laboratory practice.
Uses
Primary use: treatment of schizophrenia and related disorders. Analytical relevance arises from therapeutic drug monitoring (TDM), pharmaceutical manufacturing quality control, and occurrence studies in wastewater and aquatic environments. It may also be included in multi-residue methods covering pharmaceuticals in food-production water, sludge-amended soils, and biota.
Pharmacology & Mode of Action
Amisulpride exhibits high affinity for dopamine D2/D3 receptors. At clinical doses it antagonizes postsynaptic receptors; at low doses it can preferentially affect presynaptic autoreceptors, modulating dopaminergic transmission. Limited hepatic metabolism is reported; the parent compound can predominate in excreta, informing target-analyte selection for residue monitoring.
Regulatory Status
Amisulpride is authorized in multiple regions for treatment of schizophrenia; market authorization and conditions of use vary by jurisdiction. It is not a veterinary drug and typically has no food MRLs under pesticide/veterinary frameworks. Environmental monitoring and wastewater quality programs may include amisulpride in watch lists or investigative screening panels subject to local regulation.
Monitoring & Matrices
Typical matrices: surface water, groundwater, wastewater influent/effluent, sludge, sediment, biota, soil, and occasionally drinking water; clinical matrices (plasma/serum/urine) for TDM and pharmacokinetic studies; pharmaceutical products for QC. Method development should account for matrix effects and potential ion suppression in complex environmental extracts.
Sample Preparation
- Water: direct injection or SPE (reversed-phase or mixed-mode) with appropriate conditioning and elution; adjust pH to improve retention of basic analytes.
- Wastewater/Surface water: SPE with matrix cleanup; consider filtration and isotope-dilution to correct recovery.
- Solid matrices (sludge/soil/sediment): solvent extraction (e.g., MeOH or MeOH/water), assisted by agitation or ultrasonic extraction, followed by cleanup (dispersive SPE or cartridge SPE).
- Biota/food: QuEChERS-style extraction with acidic modifiers, followed by cleanup tailored to lipids/proteins.
- Clinical: protein precipitation (acetonitrile or methanol) and dilution; SPE for enhanced sensitivity.
Analytical Methods
- Chromatography: LC with reversed-phase columns (C18 or polar-embedded phases); mobile phases with volatile buffers (e.g., formic acid/ammonium formate) for robust ionization.
- Detection: LC–ESI(+)-MS/MS in MRM provides selectivity and sub-ng/L to low ng/mL sensitivity depending on matrix and injection volume.
- Confirmation: retention time matching, ion ratio criteria, and where applicable high-resolution MS for accurate mass confirmation.
- Carryover and adsorption mitigation: use low-adsorption plastics, appropriate wash solvents, and column/needle rinses due to basic/polar properties.
Calibration & Quality Control
- Use matrix-matched calibration or standard addition for complex matrices.
- Apply isotope-dilution with a stable isotope-labelled internal standard where available to correct for recovery and matrix effects.
- Include procedural blanks, fortified blanks, and ongoing precision and recovery (OPR) samples.
- Verification via second-source standards where required by accreditation.
Reference Materials
HPC Standards provides certified reference materials for Amisulpride as neat solids and ready-to-use solutions in high-purity solvents. On request, stable isotope-labelled derivatives and custom concentrations are available. Each lot includes a certificate with purity, uncertainty, and traceability data to support ISO/IEC 17025 and ISO 17034 workflows.
Stability & Storage
Store reference materials according to the certificate of analysis (typically refrigerated, protected from light and moisture). Prepare working solutions in suitable solvent systems and monitor stability with control charts. Avoid repeated freeze–thaw cycles; use aliquots to maintain integrity.
Health Impact – Human Toxicity
Amisulpride affects dopaminergic pathways. Reported adverse effects in therapeutic contexts can include extrapyramidal symptoms at higher doses, hyperprolactinemia, and potential QT interval prolongation. Laboratory handling should prevent exposure; classification and workplace limits depend on local regulations.
Occupational & Laboratory Safety Measures
Handle in a fume hood with appropriate PPE (lab coat, nitrile gloves, safety glasses). Avoid inhalation and skin contact. Implement spill control procedures and dispose of waste according to pharmaceutical/chemical regulations. Consult SDS for detailed hazard and first-aid guidance.
Environmental Impact
Due to limited metabolism and renal excretion in patients, amisulpride can enter wastewater systems predominantly as the parent compound. Conventional wastewater treatment may only partially remove it, leading to occurrence in effluents and receiving waters at trace concentrations. Persistence and mobility warrant targeted monitoring.
Effects on Wildlife and Aquatic Systems
As a bioactive compound, amisulpride may pose sublethal risks to aquatic organisms at sufficient exposure levels, particularly via neurotransmission pathways. Ecotoxicological thresholds vary by species; site-specific risk assessment and trend monitoring are recommended.
Regulation & Compliance
While food MRLs typically do not apply, laboratories may need to meet regulatory or guideline-based reporting limits for pharmaceuticals in water (e.g., drinking water guidance values, local watch lists). Use validated methods, documented measurement uncertainty, and traceable reference materials to demonstrate compliance.
Data Reporting & Units
Report results in matrix-appropriate units (e.g., ng/L for water, ng/g dry weight for solids, ng/mL for biological fluids). Include measurement uncertainty, recovery-corrected concentrations when applicable, LOQ/LOD, and accreditation details. Provide qualifier ion ratios and retention times for confirmatory analyses.
Applications for Industry & Laboratories
- Environmental labs: surveillance of pharmaceuticals in wastewater and surface waters.
- Contract research/manufacturing: QC and release testing of pharmaceutical products.
- Clinical and toxicology labs: method development and proficiency testing support (where permitted).
- Academic and government: occurrence studies, fate and transport, and treatment performance evaluation.
Related Compounds & Metabolites
Analytical scopes may include other substituted benzamides or antipsychotics, and potential minor metabolites of amisulpride where relevant to study objectives. Parent-compound targeting is commonly prioritized due to limited metabolism.
Documentation & Certificates
Each HPC Standards reference material is accompanied by a Certificate of Analysis detailing identity, purity, concentration (for solutions), expanded uncertainty, storage conditions, and traceability, supporting method validation, system suitability, and routine QA/QC.


