New user? / Forgot your password?
0 Item | 0,00 €
Sulfisomidine – Reference Materials for Residue Analysis
Sulfisomidine Reference Materials Precise, compliant, ready to use
Ensure confident LCMSMS quantification with HPC Standards high-purity Sulfisomidine reference materials. Manufactured to international quality requirements, our neat solids and ready-to-use solutions support method development, calibration, and routine QC in food and environmental testing. Benefit from batch-specific CoAs, traceable documentation, and expert technical supportideal for meeting MRL-driven regulatory demands and achieving robust, defensible results across complex matrices.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 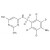 | 693335 | 1X5MG | |
ISO 17034 Certified Reference Material D4-Sulfisomidine solution | 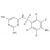 | 693336 | 1X1ML | |
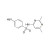 | 674888 | 1X250MG | Please log in. | |
ISO 17034 Certified Reference Material Sulfisomidine solution | 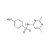 | 682401 | 1X5ML | Please log in. |
HPC Standards GmbH supplies high-quality reference materials for Sulfisomidine to support accurate residue analysis in food and environmental matrices. Our materials meet international quality requirements and are designed for regulatory compliance testing and routine monitoring.
Overview
Sulfisomidine (INN), also known as sulphasomidine (former BAN), sulfamethin, or sulfaisodimidine, is a sulfonamide antibacterial structurally related to sulfadimidine. It has been used in human and veterinary medicine; residues can occur in animal-derived foods and the environment.
Pharmacokinetics: minor acetylation; primary renal excretion (~85%). Administration: oral. Therapeutic class: systemic sulfonamide (ATC J01EB01).
Synonyms and Identifiers
IUPAC: 4-amino-N-(2,6-dimethylpyrimidin-4-yl)benzenesulfonamide
CAS: 515-64-0 | PubChem CID: 5343 | UNII: W03L3ODK6E | KEGG: D01526 | ChEBI: CHEBI:32166 | ChEMBL: 485696
Molecular formula: C12H14N4O2S | Molar mass: 278.33 g/mol
Chemical and Structural Properties
Functional class: sulfonamide; primary aromatic amine and sulfonamide group enable ionizable behavior and interactions with polar phases.
Representative structure notation (SMILES): O=S(=O)(Nc1nc(nc(c1)C)C)c2ccc(N)cc2
Uses and Applications
Antibacterial agent used historically in therapeutic contexts. In residue analysis, Sulfisomidine is a target compound in multi-residue methods for veterinary drug surveillance in meat, milk, eggs, and aquaculture, as well as in surface and wastewater.
Occurrence and Sources of Residues
Residues may result from veterinary treatments, off-label use, or cross-contamination in feed. Environmental presence can arise from excretion and insufficient wastewater removal.
Regulatory Context
Maximum residue limits (MRLs) for sulfonamides, including Sulfisomidine where applicable, are set or guided by regional frameworks (e.g., EU veterinary drug regulations, Codex, national MRL lists). Laboratories should consult the latest legal texts for matrix- and species-specific limits.
Monitoring and Compliance
Routine monitoring focuses on animal-derived foods (muscle, liver, kidney, milk, eggs, honey) and environmental waters. Confirmatory methods typically use LC–MS/MS with isotope dilution where available. Participation in proficiency testing and use of traceable reference materials support defensible results.
Analytical Methods
Instrumental: LC–MS/MS (MRM) for selectivity and sensitivity; HPLC–UV/FLD as screening where applicable. HRMS can support non-target confirmation.
Typical performance goals: low µg/kg levels in food matrices; ng/L–µg/L in water. Method validation follows SANTE/ISO/IEC 17025 and relevant veterinary drug guidelines (linearity, recovery, precision, CCα/CCβ, matrix effects).
Sample Preparation
Food: protein precipitation and solvent extraction (e.g., acidified MeOH/ACN), followed by SPE cleanup (weak anion exchange or mixed-mode) or QuEChERS adaptations.
Water: SPE preconcentration (HLB/mixed-mode) with pH control to optimize retention of ionizable sulfonamides.
Matrix Considerations
Strong matrix effects (ion suppression/enhancement) may occur in high-fat or high-protein matrices. Matrix-matched calibration and internal standards are recommended. Hydrolysis/acetyl-metabolite back-conversion should be considered during sample prep.
Quality Assurance
Use of characterized reference materials (neat and solutions), system suitability checks, bracketing calibration, procedural blanks, and recovery spikes ensure traceability and robustness.
Health Impact – Human Toxicity
Class-related effects for sulfonamides include hypersensitivity reactions and potential hematologic or dermatologic adverse events. Risk to consumers is managed via adherence to MRLs and withdrawal periods.
Environmental Impact
Sulfonamides can persist and exert selective pressure on microbial communities, potentially contributing to antimicrobial resistance development. Monitoring in effluents and surface waters supports environmental risk management.
Safety Measures and Handling
Handle Sulfisomidine reference materials in a laboratory environment using appropriate PPE (gloves, lab coat, eye protection) and engineering controls. Avoid dust formation and inhalation. Follow local chemical hygiene and waste disposal regulations.
Storage and Stability
Store sealed, protected from light and moisture, at recommended temperatures (typically 2–8 °C for solutions; cool, dry conditions for neat material). Allow for equilibration to room temperature before opening to minimize condensation. Observe expiry dates and re-test intervals.
Reference Materials from HPC Standards
HPC Standards provides Sulfisomidine reference materials for qualitative and quantitative analysis in food and environmental testing. Materials are produced and tested under international quality requirements and are suitable for method development, calibration, and quality control.
Available formats typically include neat solids and ready-to-use solutions; stable isotope‑labelled derivatives may be available to support isotope dilution LC–MS/MS.
Ordering and Support
Benefit from batch-specific CoAs, detailed documentation, and technical support for method setup and validation. For product specifications, pack sizes, and lead times, contact our sales team or consult our online catalog.


