New user? / Forgot your password?
0 Item | 0,00 €
Monalide reference materials for residue analysis
Monalide Precision Reference Materials for Confident Compliance
Elevate your residue analysis with Monalide reference materials from HPC Standards GmbH. Our high-purity neat and solution standards are engineered for LC-MSMS and complementary chromatographic workflows, delivering traceable calibration, robust method validation, and cross-lab comparability. Each lot ships with a comprehensive CoA, SDS, and uncertaintytraceability statements to support ISOIEC 17025-accredited processes and regulatory frameworks e.g., EU MRLs, Codex, national tolerances.
Why laboratories choose HPC Standards
- High-integrity Monalide standards verified by orthogonal techniques purity and identity documented on the CoA
- Ready-to-use concentrations in LC-MS compatible solvents acetonitrile, methanol, water-based mixes
- Designed for food, feed, water, soil, sediment, and biota matricesideal for calibration, QC, PT, and recovery studies
- Custom concentrations, multi-analyte mixes, and isotope-labelled derivatives available on request
- Global supply, temperature-appropriate packaging, and audit-ready documentation
Ensure reliable quantification and streamlined compliance. Partner with HPC Standards for Monalide reference materials you can trust.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 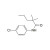 | 674310 | 1X100MG | Please log in. |
Monalide solution | 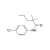 | 685079 | 1X5ML | Please log in. |
ISO 17034 Certified Reference Material | 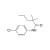 | 693861 | 1X50MG | Please log in. |
High-integrity Monalide reference materials for quantitative residue analysis in food and environmental matrices. Manufactured and tested by HPC Standards GmbH according to international quality requirements to support regulatory compliance and method validation.
Overview
Monalide is a pesticide-related compound monitored in food, feed, water, and environmental samples. Reliable quantification of Monalide residues requires traceable reference materials to establish calibration, verify instrument performance, and ensure comparability of results across laboratories.
HPC Standards GmbH provides neat and solution-based Monalide reference materials designed for LC-MS/MS workflows and other chromatographic techniques. Each lot is accompanied by comprehensive documentation (CoA, safety data, uncertainty, and traceability statements).
Typical uses
- Calibration and verification for routine monitoring programs in food safety, environmental surveillance, and industrial hygiene.
- Method development and validation (linearity, accuracy, precision, LOD/LOQ determination, recovery studies).
- Proficiency testing, interlaboratory comparability, and internal quality control (QC) samples.
Regulatory context
Monalide residues may fall under jurisdiction of regional and international frameworks (e.g., EU MRLs, EFSA guidance, Codex Alimentarius, and national tolerances such as US EPA). Laboratories require validated methods and traceable reference materials to demonstrate compliance with these requirements and accreditation standards (e.g., ISO/IEC 17025).
Monitoring and target matrices
- Food and feed: raw agricultural commodities, processed products, baby food, dietary supplements.
- Environmental: surface water, groundwater, wastewater, sediments, soil, biota.
- Industrial and research samples: formulation studies, migration/transfer assessments, degradation and metabolite profiling.
Analytical methods
- Primary: LC-MS/MS with MRM detection for selectivity and sensitivity in complex matrices.
- Complementary: LC–HRMS for non-target/suspect screening and confirmation; GC-MS/MS where analytically applicable following derivatization or specific sample preparation.
- Sample preparation: QuEChERS-based extraction for food; SPE for water; tailored clean-up (dSPE/SPE) to mitigate matrix effects. Method parameters should be optimized for recovery, matrix suppression control, and measurement uncertainty.
Health impact
Human toxicity (general): Hazard characterization for Monalide depends on dose, route, and exposure duration. Appropriate laboratory controls and PPE are required to minimize inhalation, dermal, and ingestion risks. Consult the SDS for classification, first-aid measures, and occupational exposure guidance.
Environmental impact
Fate and transport: Environmental behavior (persistence, mobility, and transformation) is compound- and matrix-dependent and should be assessed under relevant conditions.
Effects on wildlife: Potential risks to aquatic and terrestrial organisms necessitate monitoring of water bodies, sediments, and biota where Monalide may occur.
Safety measures
- Use in a fume hood with appropriate PPE (lab coat, nitrile gloves, safety glasses).
- Avoid skin contact and inhalation; prevent release to the environment.
- Follow good laboratory practice (GLP) for handling, spill response, and waste disposal in accordance with local regulations.
Product specification (HPC Standards)
- Formats: neat solid/liquid (where applicable) and ready-to-use solution reference materials at defined mass fractions or concentrations.
- Solvents (typical): acetonitrile, methanol, or water-based mixtures selected for stability and compatibility with LC-MS.
- Purity and identity: verified by orthogonal techniques (e.g., LC/UV, LC-MS, NMR where applicable); provided on the CoA with uncertainty.
- Packaging: amber glass vials with tamper-evident closures to protect from light and contamination.
Quality and traceability
- Manufactured under stringent quality control aligned with international requirements.
- Accompanied by a Certificate of Analysis detailing assigned value, measurement uncertainty, analytical methods, and traceability.
- Suitable for ISO/IEC 17025-accredited workflows and method validation studies.
Storage and stability
- Recommended storage: refrigerated or frozen (e.g., 2–8 °C or ≤ –18 °C) and protected from light; avoid repeated freeze–thaw cycles.
- Stability statements and shelf-life are lot-specific; see CoA for expiry date and handling instructions.
Method validation guidance
- Establish calibration with multi-point curves across the required range; verify linearity and matrix effects using matrix-matched standards or standard addition.
- Confirm accuracy via recovery experiments and certified control samples; evaluate precision (repeatability/reproducibility) and measurement uncertainty.
- Include system suitability, blanks, and fortified QC levels per batch.
Documentation provided
- Certificate of Analysis (value assignment, uncertainty, purity, identity confirmation, storage conditions).
- Safety Data Sheet (hazard classification, handling, PPE, transport, disposal).
- Technical datasheet (solvent system, recommended use, compatibility notes).
Related compounds and metabolites
Reference materials for structurally related pesticides, metabolites, and stable isotope-labelled derivatives may be available to support confirmation, recovery correction, and isotope-dilution quantification. Contact us for custom mixtures and isotope-labelled Monalide derivatives on request.
Custom solutions and mixtures
- Custom concentration levels and solvent systems tailored to your method.
- Multi-analyte mixes for screening and routine monitoring panels.
- Bulk and lot-reservation options for long-term studies and harmonized QC.
Logistics and compliance
- Global shipping with temperature-appropriate packaging where required.
- Transport in accordance with applicable regulations; documentation included for customs and quality audits.
Analytical standards from HPC Standards
HPC Standards GmbH specializes in the manufacture of reference materials for pesticides, veterinary drugs, their metabolites, and stable isotope-labelled derivatives. Our Monalide reference materials support accurate quantification, robust method validation, and reliable compliance testing in food and environmental analysis.


