New user? / Forgot your password?
0 Item | 0,00 €
Isoeugenol Reference Materials for Residue and Quality Analysis
Isoeugenol Reference Materials PRECISION for Regulatory-Grade Results
Ensure confident identification and quantification of isoeugenol and its cistrans isomers with high-purity reference materials from HPC Standards. Built for food, cosmetic, fragrance, and environmental testing, our materials deliver verified identity, certified purity, and traceable documentation to meet international quality requirements. Choose from neat standards and ready-to-use solutions, with isomer-specific and stable isotopelabelled options available to strengthen method robustness, isomer resolution, and matrix performance. Rely on fast global supply, custom concentrations and solvent systems, and expert method support to keep your QC programs compliant, consistent, and audit-ready.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
D3-Isoeugenol solution | 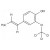 | 693996 | 1X1ML | |
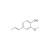 | 683790 | 1X1000MG | Please log in. |
High-precision reference materials for isoeugenol and its isomers to support reliable identification, quantification, and compliance in food, cosmetic, fragrance, and environmental testing.
Overview
Isoeugenol is a phenylpropanoid (C10H12O2) and positional isomer of eugenol, occurring naturally in essential oils such as ylang-ylang and clove derivatives. It exists as cis- and trans-isomers and is widely used as a fragrance ingredient, flavoring substance, and chemical intermediate, notably in vanillin manufacture.
Due to its sensitization potential and occurrence in consumer products and environmental matrices, isoeugenol is a routine target in regulatory compliance testing, product stewardship, and surveillance studies.
Chemical Identity and Properties
Synonyms: 2-Methoxy-4-propenylphenol (isomeric); 4-Hydroxy-3-methoxy-1-propenylbenzene. CAS: 97-54-1 (isoeugenol, mixture or unspecified isomer). Structural isomers: cis-isoeugenol and trans-isoeugenol.
Key properties: aromatic phenolic compound; moderately volatile; lipophilic; prone to air-oxidation and isomerization under light and alkaline conditions. Partitioning favors organic phases; amenable to GC and LC analysis.
Applications and Uses
Fragrance ingredient in perfumery, cosmetics, detergents, and household products; flavoring agent in food and beverages at low levels; intermediate in vanillin production via oxidative processes; research chemical in aroma and natural products chemistry.
Industrial relevance includes product formulation control, incoming raw material verification, and finished-goods quality assurance.
Regulatory Status
Cosmetics (EU): Isoeugenol is listed among fragrance allergens requiring labeling when exceeding 0.001% in leave-on products and 0.01% in rinse-off products as per EU Cosmetic Regulation. Maximum use levels are further restricted by IFRA Standards depending on product category; consult the latest IFRA Amendment for limits.
Classification and labeling: Commonly classified as a skin sensitizer under GHS/CLP. Mixture classification depends on concentration and product matrix.
Flavor safety: Evaluated as a flavoring substance by expert bodies; typically permitted at low concentrations consistent with good manufacturing practice. Always verify current regional regulations (EU, US, Codex, national authorities).
Monitoring and Compliance
Routine monitoring focuses on cosmetics, personal care, fragrances, detergents, air care products, food and beverages, essential oils, and environmental samples impacted by industrial discharges or consumer use.
Compliance documentation typically requires verified identity, purity, traceability of reference materials, validated methods, and retention of chromatographic and spectral data.
Human Toxicity and Health Effects
Primary concern: dermal sensitization leading to allergic contact dermatitis. Risk increases with concentration and repeated exposure. Eye and skin irritation may occur.
Toxicokinetics: rapid metabolism via conjugation pathways; systemic toxicity is generally low at typical consumer exposure levels but sensitization warrants strict concentration control and labeling.
Environmental Impact and Fate
Behavior: biodegradation is expected in aerobic conditions; however, acute and chronic aquatic toxicity can occur at elevated concentrations. Adsorption to organic matter may be significant.
Effects on wildlife: potential adverse effects on aquatic organisms at sufficient exposure; risk management should minimize release to wastewater and surface waters.
Safety Measures for Handling
Implement closed handling where feasible; use local exhaust ventilation. Personal protective equipment: chemical-resistant gloves, lab coat, safety goggles; avoid skin contact and inhalation of vapors.
Storage: store in amber, tightly sealed containers at cool temperature, under inert gas if possible. Protect from light, heat, and air to limit oxidation and isomerization. Segregate from strong oxidizers and bases.
Analytical Methods
GC-FID/GC-MS: primary techniques for quantification and identity; separate cis/trans isomers on polar or intermediate-polarity columns. Headspace or SPME-GC-MS suitable for volatile matrices.
LC-UV/LC-MS/MS: applicable for complex or thermally labile matrices; electrospray or APCI ionization depending on method development.
Sample preparation: solvent extraction or SPE for cosmetics and environmental waters; dilution in suitable organic solvents for fragrances and essential oils; QuEChERS-style cleanup for complex consumer products where appropriate.
Method Performance and QA/QC
Typical LOQs are in the low µg/kg to mg/kg range depending on matrix and detector. Use isomer-specific calibration where separation is achieved. Confirm identity with retention index and mass spectral matching.
QC measures: system suitability with reference solutions, matrix-matched calibration, use of surrogate or isotope-labeled internal standards, and routine verification with independent reference materials.
Sample Matrices and Typical Targets
Cosmetics and personal care: creams, lotions, shampoos, deodorants, fine fragrances. Household: detergents, fabric softeners, air fresheners. Flavor matrices: beverages, confectionery, baked goods. Natural products: essential oils, plant extracts. Environmental: influent/effluent, surface waters, sediments, indoor air (emissions from fragranced products).
Storage and Stability in Matrices
In-solution stability improves under low temperature and oxygen exclusion. Light and alkaline pH can promote isomerization and oxidative degradation. Use fresh calibration solutions regularly and verify against a primary reference material.
Reference Materials
HPC Standards provides high-purity reference materials for isoeugenol to support accurate qualitative and quantitative analysis across food, cosmetic, fragrance, and environmental applications.
Available options include neat materials and ready-to-use solutions at defined concentrations, with isomer-specific materials (cis- and trans-isoeugenol) on request. Stable isotope-labeled derivatives can be provided to enhance method robustness and traceability.
Quality and Documentation
Each reference material is supplied with a comprehensive Certificate of Analysis detailing identity confirmation (e.g., GC/LC, MS), purity, uncertainty, traceability, and storage recommendations. Batches are produced under rigorous quality controls aligned with international requirements.
Method Development Support
Support for selection of appropriate solvents, calibration ranges, isomer resolution strategies, and internal standardization. Guidance on adapting methods for challenging matrices and meeting current regulatory expectations.
Related Compounds and Interferences
Potential interferences include eugenol, methyleugenol, and structurally related phenylpropanoids present in essential oils. Adequate chromatographic resolution and mass spectrometric confirmation are recommended to avoid misidentification.
Logistics and Customization
Custom concentrations, solvent systems, ampoule sizes, and multi-analyte mixes can be prepared to streamline routine QC and high-throughput screening. Global shipping with appropriate temperature and light protection is available.


