New user? / Forgot your password?
0 Item | 0,00 €
Bis(4-nitrophenyl)urea — Reference Materials and Technical Information
Bis4-nitrophenylurea High-Purity Reference Materials for Confident LC-Based Quantification
Accelerate your QAQC with Bis4-nitrophenylurea standards from HPC Standards. Our high-purity, fully characterized reference materialsavailable as neat solids and custom solution blendsenable precise identification, calibration, and method validation across industrial, environmental, and research matrices. Delivered with CoA, SDS, and technical guidance, they support ISOIEC 17025 and ISO 17034 principles, ensuring traceable, defensible results and regulatory compliance. Optimize LC-UVDAD or LCMSMS workflows with matrix-appropriate formats, custom concentrations, and multi-analyte mixes tailored to your monitoring programs.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 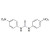 | 689015 | 1X250MG | Please log in. |
High-purity reference materials for precise identification, quantification, and method validation of Bis(4-nitrophenyl)urea across industrial, environmental, and research matrices. Manufactured and tested to international quality requirements to support regulatory compliance and robust QA/QC.
Overview
Bis(4-nitrophenyl)urea is an aromatic urea bearing two para-nitrophenyl substituents. It is a hydrogen-bond donor/acceptor capable compound, often used in research and industrial contexts where nitroaromatic ureas are relevant as functional materials, auxiliaries, or transformation products. Due to its low volatility and polar functionalities, liquid chromatography-based methods are typically favored for analysis.
In residue and contaminants monitoring programs, Bis(4-nitrophenyl)urea may be investigated as a process-related impurity, by-product, or environmental contaminant in specific industrial scenarios. Reliable quantification requires well-characterized reference materials to ensure traceability and comparability of results.
Chemical Identity and Synonyms
Preferred name: Bis(4-nitrophenyl)urea; also written as N,N′-bis(4-nitrophenyl)urea or 1,3-bis(4-nitrophenyl)urea.
General formula: C13H10N4O5 (aromatic urea with two p-nitrophenyl groups).
Structural features: urea core (–NH–CO–NH–) linking two para-nitrophenyl rings bearing nitro (–NO2) substituents.
Physicochemical Properties
Appearance: typically a pale yellow to yellow crystalline solid (nitroaromatic character).
Solubility: low in water; higher solubility in polar organic solvents (e.g., DMSO, DMF, acetonitrile) due to hydrogen-bonding interactions; limited volatility (non-amenable to direct GC without derivatization).
Chemical behavior: strong hydrogen-bond donor (urea NH) and acceptor (carbonyl, nitro oxygen) sites; thermally stable relative to many organics but subject to decomposition at elevated temperatures; light-sensitive nitroaromatic moieties may warrant protection from light for long-term stability.
Uses and Application Areas
- Research chemical for studies involving urea-based hydrogen-bond donors and supramolecular interactions.
- Potential auxiliary or intermediate in specialty chemicals and materials workflows.
- Target/analyte in industrial hygiene, product stewardship, and environmental screening where nitroaromatic ureas are relevant.
Regulatory Status
Regulatory coverage depends on jurisdiction and intended use (industrial chemical vs. laboratory reagent). While not a registered pesticide active substance, it may fall under chemical control frameworks (e.g., REACH-like inventories, workplace exposure regulations) and sector-specific product safety rules.
Analytical monitoring may be requested within supply-chain due diligence, product compliance checks, or site-specific environmental permits. Use laboratory-grade reference materials to ensure defensible reporting against internal or external specifications.
Monitoring and Target Matrices
- Industrial process streams: reaction mixtures, intermediates, and final product quality control.
- Environmental: wastewater, surface water near industrial sites, soils and sediments.
- Materials and consumer products (as applicable): polymer matrices, coatings, technical articles (screening for residuals/by-products).
- Laboratory research samples: synthesis verification, mechanistic studies, and stability testing.
Health Impact — Human Toxicity
- Nitroaromatic compounds can present systemic toxicity risks; handle as harmful if inhaled, ingested, or in contact with skin and eyes.
- Potential irritant effects to skin, eyes, and respiratory tract; minimize dust and aerosol formation.
- Follow institutional exposure control strategies (engineering controls, PPE) and consult the SDS for current hazard classification and first-aid measures.
Environmental Impact — Effects on Wildlife and Ecosystems
- Nitroaromatic structures may be persistent and exhibit toxicity to aquatic organisms; avoid uncontrolled releases to the environment.
- Implement appropriate waste handling and effluent treatment according to local regulations; consider monitoring in receiving waters if relevant to site operations.
Safety Measures — Handling and Storage
- Handle in a fume hood with suitable PPE: lab coat, safety glasses, and chemically resistant gloves; use dust control practices.
- Storage: tightly closed in original container, dry, and protected from light; recommended 2–8 °C for long-term stability unless otherwise stated on the certificate.
- Segregate from strong oxidizers and reducing agents; follow good housekeeping to prevent contamination of other analytes and standards.
Transport and Classification
Transport classification depends on current SDS and local implementation of GHS/UN rules. Package to prevent breakage, minimize exposure, and maintain temperature/light protection as indicated.
Analytical Methods
- LC-UV/LC-DAD: suitable due to aromatic chromophores; optimize wavelength in the near-UV range.
- LC-MS/MS: preferred for selectivity and sensitivity in complex matrices; negative or positive ionization to be evaluated empirically.
- GC-MS: generally requires derivatization due to low volatility and thermal lability.
- Orthogonal confirmation: NMR and FTIR for identity; HRMS for accurate mass confirmation.
Typical parameters: reversed-phase LC with C18 phase, water–acetonitrile or water–methanol gradients; acidic modifiers to improve peak shape; column temperature control to improve reproducibility.
Sample Preparation
- Aqueous matrices: SPE (e.g., polymeric reversed-phase) with solvent elution; evaluate recovery vs. pH and ionic strength.
- Solid matrices: solvent extraction (acetonitrile, acetone, or mixtures) with sonication or shaker-assisted protocols; cleanup via SPE or dispersive sorbents to reduce matrix effects.
- Materials/polymers: exhaustive extraction or targeted solvent swelling based on polymer chemistry; validate extraction efficiency and potential analyte adsorption.
Reference Materials from HPC Standards
HPC Standards provides Bis(4-nitrophenyl)urea reference materials as neat solids and custom solution blends to support identification, calibration, and quality control.
Key features:
- High chemical purity with full characterization (LC/GC purity profile, identity confirmation).
- Traceability and uncertainty budgets suitable for accredited laboratories (ISO/IEC 17025 and, where applicable, ISO 17034 principles).
- Custom concentrations, solvent systems, and multi-analyte mixes on request for method development and routine monitoring.
Stability, Shelf Life, and Commutability
- Supplied with recommended storage conditions and shelf-life statement; stability monitored under controlled conditions.
- Packaged in amber glass to mitigate photolysis; inert gas headspace used where appropriate.
- Commutable across LC-UV and LC-MS/MS workflows when stored and handled as directed; re-verify calibration on receipt and at defined intervals.
Quality Assurance and Compliance
- Delivered with Certificate of Analysis detailing purity, identity, lot number, preparation date, uncertainty (for solutions), and storage instructions.
- Supports GLP/GMP-compatible documentation needs and audit trails in accredited laboratories.
- Batch-to-batch consistency ensures reliable long-term monitoring programs.
Method Validation and Calibration Guidance
- Establish calibration ranges that bracket expected concentrations; use matrix-matched calibration or standard addition for complex matrices.
- Verify linearity, accuracy (recovery), precision (intra-/inter-day), LOD/LOQ, selectivity, and robustness per ISO 17025/ICH/Q2(R2)-style guidance.
- Incorporate independent QC levels (low/mid/high) and system suitability checkpoints for retention time, peak shape, and response stability.
Documentation Provided
- Certificate of Analysis (CoA) with analyte identity and purity data.
- Safety Data Sheet (SDS) with hazard classification and handling guidance.
- Technical datasheet with recommended analytical conditions and storage notes (where applicable).
Ordering Information and Custom Solutions
- Formats: neat material and ready-to-use solutions at defined concentrations in acetonitrile, methanol, or custom solvents.
- Packaging sizes suitable for method development, routine QC, and proficiency testing support.
- Custom mixtures with related nitroaromatics or process markers available on request to streamline multi-residue workflows.


