New user? / Forgot your password?
0 Item | 0,00 €
Monepantel Reference Materials
Monepantel Precision Reference Materials for Confident Residue Analysis
Accelerate your veterinary drug surveillance with high-purity Monepantel reference materials from HPC Standards. Designed for food and environmental laboratories, our traceable standards and ready-to-use solutions enable accurate LC-MSMS quantification across muscle, liver, kidney, fat, milk, soil, and watersupporting method development, calibration, and ISO-compliant QC. Each lot is documented for purity, uncertainty, stability, and storage to meet international quality requirements. Optional stable isotopelabelled analogues enhance accuracy and robustness, ensuring reliable results below MRLs and seamless audit readiness. Choose HPC Standards as your partner for dependable calibration, regulatory compliance, and consistent performance.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 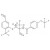 | 692703 | 1X5MG | Please log in. |
D5-Monepantel solution | 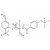 | 693787 | 1X1ML | Please log in. |
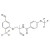 | 677135 | 1X10MG | Please log in. | |
Monepantel solution | 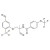 | 693788 | 1X1ML | Please log in. |
High-quality reference materials for confident quantification of monepantel and related analytes in food and environmental matrices to ensure regulatory compliance and robust residue monitoring.
Overview
Monepantel is a broad-spectrum anthelmintic of the amino-acetonitrile derivatives class used primarily in small ruminants. It targets nematode-specific nicotinic acetylcholine receptor subtypes, providing efficacy against strains resistant to older drug classes. For laboratories, validated reference materials are essential to develop, calibrate, and verify analytical methods for residue determination in compliance programs.
HPC Standards provides reference materials for monepantel tailored to the needs of food and environmental analysis, tested according to international quality requirements and suited for routine and confirmatory methods.
Uses in Veterinary Medicine
Indicated for the control and treatment of gastrointestinal nematodes in sheep and other small ruminants. Applied mainly as an oral drench on farm. Its spectrum includes key genera implicated in production losses and parasite resistance management programs.
Use is subject to veterinary supervision, adherence to label restrictions, and observance of withdrawal periods prior to slaughter or milk collection.
Mode of Action
Monepantel modulates nematode-specific nicotinic acetylcholine receptor subunits, leading to paralysis and death of susceptible helminths. The selectivity toward parasite receptors underpins a favorable mammalian safety profile relative to older classes.
Resistance can emerge under selection pressure; integrated parasite management and rotation strategies are recommended to preserve efficacy.
Regulatory Status and MRLs
Monepantel is an approved veterinary medicinal substance in several jurisdictions for use in food-producing animals. Maximum residue limits and withdrawal periods may be established for edible tissues and milk depending on region.
Laboratories should verify current national and regional regulations and MRLs set by competent authorities and align method limits of quantification accordingly.
Monitoring and Compliance
Residue monitoring focuses on edible tissues such as muscle, liver, kidney, and fat, as well as milk where applicable. Surveillance programs may also include feed, water, manure, and environmental matrices adjacent to treated herds.
Accurate calibration using traceable reference materials is critical for screening, quantitation, and confirmatory analysis to support regulatory decisions and quality assurance.
Target Matrices
Food: ruminant muscle, liver, kidney, fat, milk.
Environmental: soil and manure-amended soil, run-off water, sediment, surface water near livestock operations.
Sample Preparation
Common approaches include protein precipitation followed by liquid-liquid extraction or solid-phase extraction clean-up tailored to matrix fat content. Partitioning with acetonitrile and dispersive clean-up can mitigate matrix effects in complex tissues.
Method optimization should address recoveries across fortified levels, matrix-matched calibration, and stability of analytes during extraction and storage.
Analytical Methods
LC-MS/MS is the primary technique for selective and sensitive quantitation of monepantel residues, employing reversed-phase chromatography and multiple reaction monitoring. HRMS may be used for confirmatory identification.
Key parameters: specificity against isobaric interferences, linearity over regulatory ranges, limits of detection and quantification below applicable MRLs, and robustness across matrices. Isotope-dilution strategies improve accuracy and compensate for matrix effects.
Quality Assurance and Traceability
Use of traceable reference materials with stated purity, uncertainty, and stability supports ISO-compliant method validation and routine QC. Implement ongoing system suitability checks, recovery controls, and participation in proficiency testing where available.
HPC Standards products meet stringent industrial and international quality requirements to underpin reliable calibration and verification.
Health Impact (Human Toxicity)
Human exposure is expected to be low under normal dietary conditions when residues comply with established limits. Available data indicate low acute toxicity to mammals, consistent with the substance’s selective action on nematode receptors.
Risk assessments by authorities determine acceptable daily intakes and MRLs; adherence to these limits minimizes consumer risk. Laboratory handling should still treat the substance as potentially hazardous.
Occupational Safety Measures
In laboratories, handle monepantel reference materials using standard chemical hygiene: wear gloves, lab coat, and eye protection; work in a fume hood when preparing stock solutions; avoid inhalation and ingestion; prevent skin contact.
For field and farm use, follow product labels, avoid exposure during drenching, and observe withdrawal periods. Dispose of waste according to local regulations.
Environmental Impact
Post-treatment excretion can introduce residues to manure and soil. Environmental fate depends on sorption, degradation, and potential mobility. Monitoring near intensive livestock areas may be warranted to evaluate run-off to surface waters.
Good agricultural practices and manure management reduce environmental loading and off-site transport.
Effects on Wildlife and Non-target Organisms
While designed for nematode control in target hosts, exposure of non-target organisms may occur via manure-amended soils and water bodies. Sensitivity varies by species; prudent use and containment of wastes reduce ecological risk.
Storage and Stability
Store reference materials in original, tightly closed containers, protected from light and moisture, at recommended temperatures. Prepare solutions with suitable solvents and keep at low temperature to limit degradation.
Document opening dates, monitor for precipitates or discoloration, and follow expiration or re-test intervals provided on certificates.
Cross-Contamination Control
Prevent carryover by segregating standards and samples, using dedicated glassware and consumables, and implementing rigorous rinse protocols on LC-MS/MS systems. Include procedural blanks to verify cleanliness.
Related Compounds and Metabolites
Consider inclusion of major transformation products or metabolites in method scope as required by regulatory guidance. Reference materials for these analytes facilitate broader screening and confirmatory workflows.
Product Portfolio: Reference Materials for Monepantel
Available as neat materials and ready-to-use solutions at defined concentrations with documentation of purity and uncertainty. Stable isotope-labelled analogues may be offered to support isotope-dilution quantification and improve method accuracy.
Each lot is supplied with a certificate detailing characterization, recommended storage, and traceability information to support accreditation and audits.
Applications for Food and Environmental Laboratories
Suitable for method development, calibration curves, matrix-matched standards, recovery studies, quality control samples, and proficiency testing preparations across tissue and environmental matrices.
Supports both routine surveillance and targeted investigations in compliance with regulatory programs.
Why HPC Standards
Specialized manufacturer and distributor of reference materials for veterinary drugs, pesticides, metabolites, and stable isotope-labelled derivatives. Our products are tested according to international quality requirements and meet high industrial standards, enabling precise and reliable residue analysis for regulatory compliance.


