New user? / Forgot your password?
0 Item | 0,00 €
Olmesartan — Reference Materials for Residue and Environmental Analysis
Olmesartan Precision Reference Materials for Confident LCMSMS Quantification
Achieve reliable, reproducible results with HPC Standards highpurity Olmesartan reference materials. Engineered for pharmaceutical QC and environmental monitoring, our standards deliver
- SI-traceable calibration and batch-specific CoA for audit-ready compliance ISOIEC 17025, ICH Q2
- Optimized performance for LCMSMS workflows ESI, including isotope-labelled options
- Flexible formats neat and solution standards, custom mixes, matrix-matched materials
Partner with HPC Standards GmbH to streamline method development, validation, and routine surveillanceso your lab meets regulatory limits with confidence and efficiency.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Certified Reference Material Olmesartan solution | 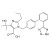 | 694572 | 1X5ML | Please log in. |
Olmesartan solution | 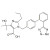 | 694573 | 1X10ML | Please log in. |
HPC Standards GmbH supplies high-purity reference materials for Olmesartan to support accurate quantification in pharmaceutical quality control and environmental monitoring. All materials are manufactured under stringent quality systems, delivered with full documentation, and optimized for LC–MS/MS workflows.
Overview
Olmesartan is an angiotensin II receptor blocker (ARB) used as an antihypertensive active pharmaceutical ingredient (API). In analytical contexts, Olmesartan (the active acid) is encountered as a parent compound in pharmaceutical products and as a transformation or emission residue in water systems. Reliable reference materials are essential for method development, validation, routine QC, and surveillance of pharmaceutical residues in environmental matrices.
Chemical Identity and Properties
Common name: Olmesartan (active acid; not the medoxomil prodrug)
Chemical class: Biphenyl tetrazole ARB (angiotensin II AT1 receptor antagonist)
General properties relevant to analysis: moderately lipophilic, acidic functionality (favors ESI negative ionization), limited volatility; stable as solid; reference solutions typically prepared in MeOH or ACN with water.
Uses
- Active ingredient in antihypertensive medications (ARB class).
- Analytical target in pharmaceutical QC (assay, purity, impurities) and stability studies.
- Environmental monitoring marker for pharmaceutical residues in wastewater, surface water, and occasionally drinking water studies.
Regulatory Landscape
- Pharmaceutical QC: compliance with ICH guidelines (e.g., assay accuracy, impurity profiling, method validation parameters such as specificity, precision, LOD/LOQ, linearity, robustness).
- Pharmacopoeial context: monographs and general chapters may guide identity, purity, and assay requirements for ARB drug substances and products; consult the applicable pharmacopeia for current specifications.
- Environment: jurisdictions increasingly request monitoring of pharmaceuticals in wastewater effluents and receiving waters; limit values are matrix- and region-specific and often study- or permit-driven rather than universal.
Monitoring and Target Matrices
- Matrices: raw and treated wastewater, surface water, groundwater, drinking water, sludge, and occasionally sediments.
- Typical concentration ranges: ng/L to low µg/L in environmental waters; µg/g levels in solid matrices (sludge/sediment) depending on source terms.
- Study designs: influent/effluent treatment performance, seasonal loads, fate and transport, and risk screening programs.
Sample Preparation and Analytical Methods
- Extraction: solid-phase extraction (SPE) using reversed-phase or mixed-mode cartridges; optimize pH to retain acidic analytes; include isotope-labelled internal standards to correct matrix effects.
- Chromatography: reversed-phase HPLC/UPLC with acidified aqueous/organic gradients; columns with high retention for polar-acidic pharmaceuticals reduce breakthrough.
- Detection: LC–MS/MS with ESI in negative ion mode is commonly used for Olmesartan; multiple reaction monitoring (MRM) transitions selected during method development.
- Validation: follow ISO/IEC 17025 and ICH Q2(R1) (or successor) for LOD/LOQ, accuracy, precision, recovery, matrix effects, and measurement uncertainty.
Calibration and Traceability
- Use gravimetrically prepared calibration solutions from certified reference materials to ensure SI traceability.
- Prefer isotope-dilution where available (stable isotope-labelled Olmesartan) to mitigate matrix effects and improve quantification accuracy.
- Apply matrix-matched calibration for complex samples (wastewater, sludge) and bracket runs with continuing calibration verification standards.
Health Impact — Human Toxicity
- Pharmacology: selective AT1 receptor antagonism reduces vasoconstriction and aldosterone effects, lowering blood pressure.
- Toxicity profile (occupational relevance): avoid ingestion, inhalation of powders, and skin/eye contact. Potential systemic effects reflect pharmacological action. Handle under good laboratory hygiene with appropriate engineering controls.
- No therapeutic claims are implied; this information is provided to inform safe handling during analytical work.
Environmental Impact
- Presence: detected in wastewater effluents and occasionally in receiving waters where pharmaceutical loads are significant.
- Fate: transformation via biodegradation, photolysis, and advanced treatment varies by system; parent Olmesartan may exhibit moderate persistence in conventional treatment.
- Bioaccumulation: expected to be low to moderate; assess site-specifically.
Effects on Wildlife and Ecosystems
- Potential risks arise from chronic exposure to low concentrations; effects are compound- and species-specific. Current ecological data for Olmesartan are limited; risk assessments typically use conservative screening benchmarks.
- Monitoring data combined with toxicity reference values support local risk characterization.
Safety Measures and Handling
- Use PPE: lab coat, nitrile gloves, safety glasses; handle powders in a fume hood or ventilated enclosure.
- Avoid dust generation; prevent inhalation and contact. Wash thoroughly after handling. Follow site-specific SOPs and SDS instructions.
- Spill response: contain, avoid release to drains, collect with inert absorbent, and dispose according to regulations.
Storage and Stability
- Solid reference materials: store tightly closed in a cool, dry, and dark place; typical long-term storage at 2–8 °C or below as indicated on the CoA.
- Reference solutions: prepare in suitable solvents (e.g., MeOH/ACN with water); store in amber vials at 2–8 °C or ≤ −20 °C; monitor stability and replace before expiry.
- Avoid repeated freeze–thaw cycles; aliquot as needed. Protect from moisture and light.
Analytical Standards
Our portfolio includes neat and solution-based reference materials for Olmesartan designed for LC–MS/MS and HPLC applications. Stable isotope-labelled internal standards may be available to support isotope-dilution quantification. Each item is supported by a detailed Certificate of Analysis.
Quality and Documentation
- Manufactured under accredited quality systems with full metrological traceability where applicable.
- Supplied with Certificate of Analysis (purity, identity, assay method, uncertainty, traceability), Safety Data Sheet, and recommended handling/storage instructions.
- Batch-specific data support method validation, system suitability, and measurement uncertainty budgets.
Applications and Industries
- Pharmaceutical manufacturers, CMOs, and CROs: assay, impurity profiling, stability studies, cleaning verification.
- Water and environmental laboratories: wastewater surveillance, surface water screening, advanced treatment performance studies.
- Regulatory and academic laboratories: occurrence studies, fate/transport research, and risk screening.
Method Development Tips
- Optimize SPE pH and wash steps to balance recovery and cleanliness.
- Use negative ESI with appropriate mobile phase modifiers to enhance sensitivity for acidic analytes.
- Apply isotope-labelled internal standards to correct for ion suppression and extraction variability.
Compliance and Reporting
- Align validation and reporting with ISO/IEC 17025, ICH Q2(R1) (or successor), and applicable regional environmental monitoring guidance.
- Document calibration traceability, measurement uncertainty, and QA/QC checks (blanks, spikes, duplicates, CCVs) in final reports.
Ordering Information
- Available pack sizes and concentrations tailored for routine QC and high-throughput monitoring.
- Custom mixtures, matrix-matched materials, and isotope-labelled variants available on request.
- Technical support from our application specialists for method setup, validation strategy, and troubleshooting.
Related Compounds
- Olmesartan medoxomil (prodrug; relevant for pharmaceutical QC).
- Phase II conjugates (e.g., glucuronides) may be relevant in environmental or metabolite studies; inquire for availability.


