New user? / Forgot your password?
0 Item | 0,00 €
Tribromoacetic acid (2,2,2-tribromoacetic acid)
Tribromoacetic acid High-Purity Reference Materials for Confident DBP and HAA Analysis
Accelerate reliable quantification with HPC Standards tribromoacetic acid 2,2,2-tribromoacetic acid reference materials. Engineered for method development, routine monitoring, and regulatory-grade QAQC, our neat and ready-to-use solutions support LC-MSMS and GC workflows in drinking water, environmental, and food-related matrices. Each lot is backed by a Certificate of Analysis, stringent purity verification, and international quality compliancedelivering traceable calibration, reproducible recoveries, and audit-ready documentation for accredited laboratories. Choose HPC Standards to strengthen your extended HAA panels e.g., HAA9HAA6Br, streamline validation, and protect data integrity.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 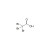 | 684089 | 1X250MG | Please log in. |
High-purity reference materials for tribromoacetic acid to support robust method development, routine monitoring, and regulatory compliance in food, water, and environmental analysis.
Overview
Tribromoacetic acid is a halogenated acetic acid used as a reference compound in the analysis of disinfection by-products (DBPs) and other environmental matrices. As a strong organic acid bearing three bromine atoms, it is relevant for laboratories monitoring haloacetic acids (HAAs) in drinking water and investigating brominated transformation products in environmental and food-related studies.
HPC Standards GmbH provides high-purity tribromoacetic acid reference materials to ensure accurate quantification, method validation, and quality control across diverse analytical workflows.
Chemical Identity
IUPAC name: 2,2,2-Tribromoacetic acid
Synonyms: Tribromoacetic acid; TBA
Chemical formula: C2HBr3O2
Substance class: Haloacetic acid (brominated)
CAS number: 75-96-7
Uses
- Reference compound for method development and calibration in HAA and DBP monitoring programs.
- Analytical control for studies on brominated transformation products in water treatment and environmental fate.
- Reagent and benchmark analyte in advanced oxidation, photolysis, and chlorination/bromination research.
Formation and Occurrence
- May form as a by-product during water disinfection when bromide is present and oxidants (e.g., chlorine, ozone) react with natural organic matter.
- Can appear as a transformation product in laboratory studies of brominated organics and during oxidative processing of environmental samples.
- Typically monitored alongside other haloacetic acids to characterize DBP profiles in drinking water and related matrices.
Regulatory and Guidelines
- Haloacetic acids are regulated as groups in several jurisdictions (e.g., HAA5 in US drinking water rules). Tribromoacetic acid is not part of HAA5 but is included in extended monitoring sets (e.g., HAA9/HAA6Br) used by utilities, researchers, and some authorities for broader DBP assessment.
- Laboratories may include tribromoacetic acid in surveillance programs, method performance studies, or risk evaluations where brominated DBPs are of interest. Always consult current local regulations and guidance for applicable limits and reporting requirements.
Monitoring and Sampling
- Matrix scope: drinking water, surface and ground waters, process waters, wastewater effluents; occasionally food or environmental extracts in transformation studies.
- Sample handling: minimize residual disinfectant (chemical quenchers as appropriate), keep samples cold, and preserve at acidic pH where method protocols require. Avoid contamination from halogenated reagents and labware.
- Quality assurance: apply internal/surrogate standards, perform field blanks, and verify recoveries across anticipated concentration ranges.
Analytical Methods
- LC-MS/MS (negative ESI): direct analysis of underivatized haloacetic acids with appropriate chromatographic retention and ion-pair or HILIC strategies. Suitable for multi-analyte HAA panels, including tribromoacetic acid.
- GC-ECD or GC-MS: typically after derivatization (e.g., esterification to methyl esters) to improve volatility and detectability. Common in routine HAA methods where sensitivity and selectivity are required.
- Calibration and QC: use matrix-matched calibration or standard addition when matrix effects are significant; employ isotopically labeled surrogates when available to correct for recovery and ion suppression/enhancement.
Health Impact
- Human toxicity: tribromoacetic acid is a strong, corrosive organic acid; contact can cause skin and eye irritation or burns, and inhalation/ingestion may be harmful. As a brominated HAA, systemic effects have been reported for related compounds in experimental studies, though data for tribromoacetic acid are comparatively limited. Apply precautionary exposure controls.
- Occupational exposure: prevent aerosol or dust formation; ensure engineering controls and appropriate PPE during handling and sample preparation.
Environmental Impact
- Persistence and mobility: haloacetic acids are water-soluble and can persist in aqueous environments, depending on conditions (pH, sunlight, microbial activity).
- Effects on wildlife: brominated HAAs may be toxic to aquatic organisms at sufficiently high concentrations. Environmental risk depends on local formation, dilution, and degradation pathways.
Safety Measures and Handling
- PPE: chemical-resistant gloves, lab coat, eye/face protection; use fume hood for weighing and solution preparation.
- First aid: in case of contact, rinse affected area with water for at least 15 minutes and seek medical attention; see product SDS for details.
- Spill management: contain, neutralize cautiously if appropriate, and collect according to institutional procedures.
Storage and Stability
- Store in a tightly closed container, in a cool, dry place, protected from moisture and incompatible materials (strong bases, strong oxidizers/reducers incompatible with halogenated organics).
- For solution reference materials, follow label instructions on solvent, concentration, and temperature; minimize freeze–thaw cycles; note expiry and requalification schedules per certificate.
Analytical Standards (Reference Materials)
HPC Standards offers tribromoacetic acid reference materials designed for residue analysis and DBP monitoring:
- Formats: neat material and ready-to-use solutions at application-relevant concentrations.
- Documentation: Certificate of Analysis (purity, identity, uncertainty where applicable), safety data sheet, and method guidance notes.
- Quality: tested according to international quality requirements to meet the highest industrial standards, supporting traceable calibration and rigorous QC in accredited laboratories.
Method Development Guidance
- Choose LC-MS/MS for minimal sample preparation and panel coverage; optimize ionization and mobile phase for brominated acids.
- For GC methods, apply validated derivatization (e.g., diazomethane or alternative methylation) and verify conversion efficiency and stability of esters.
- Assess recoveries across representative matrices; include blanks and spikes; verify linearity and measurement uncertainty with HPC Standards reference materials.
Applications
- Drinking water DBP surveillance (extended HAA panels).
- Process optimization in water treatment (bromide management, oxidant selection).
- Environmental fate and transformation studies of brominated organics.
- Interlaboratory comparisons, proficiency testing, and routine QA/QC.
Related Analytes
- Other haloacetic acids: monobromoacetic acid, dibromoacetic acid, monochloroacetic acid, dichloroacetic acid, trichloroacetic acid, tribromoacetic acid esters (derivatization products).
- Complementary DBPs: bromate, bromoform, brominated haloacetonitriles (for comprehensive DBP profiling).
Why HPC Standards
We specialize in reference materials for pesticides, veterinary drugs, their metabolites, and stable isotope-labeled derivatives. Our tribromoacetic acid reference materials support high-confidence quantification, enabling laboratories in food and environmental analysis to validate methods, meet regulatory expectations, and ensure data integrity.


