New user? / Forgot your password?
0 Item | 0,00 €
Ornidazole Reference Materials for Residue and Environmental Analysis
Ornidazole High-Purity Reference Materials for CONFIDENT RESULTS
Achieve reliable quantification across food, feed, and environmental matrices with Ornidazole reference materials from HPC Standards GmbH. Manufactured and tested to international quality requirements, our high-purity neat materials and certified solutions support LCMSMS method development, validation, and routine QCmeeting the stringent needs of accredited laboratories. Benefit from batch traceability, detailed Certificates of Analysis, and optional stable isotope-labelled derivatives for robust matrix compensation. Choose HPC Standards as your specialist partner for compliant, precise Ornidazole residue analysis.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 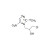 | 680037 | 1X10MG | Please log in. |
13C2,15N2-Ornidazole solution | 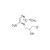 | 690931 | 1X1ML | Please log in. |
ISO 17034 Reference Material | 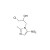 | 681579 | 1X100MG | Please log in. |
ISO 17034 Certified Reference Material | 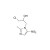 | 694393 | 1X50MG | Please log in. |
High-purity Ornidazole reference materials for precise quantification in food, feed, and environmental matrices, manufactured and tested to international quality requirements.
Overview
Ornidazole is a 5-nitroimidazole antimicrobial used primarily as an antiprotozoal and anti-anaerobic agent. In residue and environmental analysis, Ornidazole is monitored as a potential contaminant in water, wastewater, and—where applicable—animal-derived matrices. Reliable reference materials are essential for method development, validation, and routine QC.
Key identifiers: CAS 16773-42-5; antimicrobial class: nitroimidazole derivative; typical analytical determination by LC–MS/MS.
Uses
- Therapeutic agent against protozoal infections (e.g., amoebiasis, giardiasis, trichomoniasis) and certain anaerobic bacterial infections in human medicine; usage patterns may drive environmental occurrence via excretion and incomplete removal in wastewater treatment.
- In some regions, off-label or historical veterinary applications have been reported; regulatory authorizations vary by jurisdiction.
Regulatory Status
- European Union: Nitroimidazoles are generally subject to strict control in food-producing animals. Ornidazole is not authorized for use in food-producing species in the EU; no MRL is established under Commission Regulation (EU) No 37/2010, implying a zero-tolerance policy for residues in food of animal origin.
- Method performance and validation for official control of residues of veterinary medicinal products should follow Commission Implementing Regulation (EU) 2021/808 (criteria for specificity, CCα/CCβ, trueness, precision, and measurement uncertainty).
- Outside the EU, national regulations may differ; laboratories should verify local MRLs/permission status and applicable guidance documents (e.g., Codex, US, or regional frameworks).
Monitoring and Analytical Methods
- Target matrices: surface water, wastewater effluent/influent, groundwater, drinking water, sediments; where relevant, milk, eggs, honey, meat, and fish products for residue control.
- Sample preparation: SPE (reversed-phase or mixed-mode) for water; QuEChERS-based or protein-precipitation/SPE workflows for animal-origin matrices. Matrix effects are common; stable-isotope internal standards are recommended.
- Instrumentation: LC–MS/MS (MRM) is preferred for sensitivity and selectivity; HPLC–UV may be suitable for high-level screening but lacks confirmatory power. Typical LOQs are in the low ng/L for waters and ≤1–10 μg/kg for food matrices, depending on method performance.
- Quality assurance: Include fortified QC samples, procedural blanks, and matrix-matched calibration or standard addition. Use isotopically labelled internal standards when available to correct for recovery and ion suppression/enhancement.
Human Health Impact
- Pharmacological activity: Antiprotozoal/anti-anaerobic mechanism via nitroreduction and subsequent DNA-damaging intermediates.
- Toxicological profile: Can cause dose-dependent adverse effects such as gastrointestinal disturbances, CNS effects (e.g., dizziness, headache), and rare neurotoxic reactions; potential interactions with alcohol and certain drugs should be considered in clinical contexts. Data on genotoxicity/carcinogenicity are limited for Ornidazole; risk assessments should be jurisdiction-specific.
Environmental Impact
- Occurrence: Detected in municipal wastewater and impacted surface waters due to incomplete metabolism and partial removal in WWTPs.
- Fate and behavior: Nitroimidazoles may exhibit moderate persistence; biodegradation and photolysis can contribute to attenuation, but rates are matrix- and light-dependent. Sorption to sediments is variable.
- Effects on wildlife: Potential sublethal effects on aquatic microorganisms and selection pressure for antimicrobial resistance. Chronic toxicity data for higher trophic levels are limited; precautionary monitoring is advised.
Safety Measures for Laboratory Handling
- Handle Ornidazole powders and solutions in a chemical fume hood; avoid inhalation and skin/eye contact. Wear lab coat, nitrile gloves, and safety glasses.
- Storage: Keep in tightly closed containers, protected from light and moisture. Typical storage 2–8 °C for solutions; follow certificate recommendations for stability and shelf life.
- Disposal: Treat solutions and residues as pharmaceutical/organic hazardous waste per institutional and local regulations.
Method Validation and Compliance
- Validate methods in accordance with EU 2021/808 (veterinary residues) or relevant national/ISO guidelines (e.g., ISO/IEC 17025). Establish linearity, selectivity, trueness, precision, recovery, matrix effects, and measurement uncertainty.
- Use system suitability and ongoing QC (control charts, proficiency testing) to maintain data integrity. Define decision limits CCα and detection capability CCβ for confirmatory methods.
Cross-Analyte Considerations
- Co-occurrence: Other nitroimidazoles (e.g., metronidazole, dimetridazole) and their metabolites may be present; consider multi-residue LC–MS/MS methods with class-specific transitions.
- Metabolites/transformations: Reduction and conjugation products can appear in biotic matrices and during wastewater treatment; include target/ suspect screening where required.
Analytical Standards
- HPC Standards GmbH supplies Ornidazole reference materials for quantitative analysis in food and environmental applications. Products are manufactured and tested according to international quality requirements and meet the highest industrial standards.
- Available formats: neat materials and certified solutions at common concentrations (e.g., 100–1,000 μg/mL) in LC–MS grade solvents; matrix-matched calibrants available upon request.
- Isotope-labelled derivatives: Stable isotope-labelled internal standards (subject to availability) can be provided to enhance accuracy and correct for matrix effects. Please inquire for Ornidazole-labelled analogs.
- Documentation: Each lot is supplied with a Certificate of Analysis detailing identity confirmation, purity, concentration (for solutions), uncertainty, solvent, storage, and expiry. Batch traceability and homogeneity testing support regulatory compliance.
- Custom services: Tailored mixes (multi-analyte nitroimidazole panels), alternative solvents/concentrations, and large-volume batches for routine monitoring programs.
Applications in Food and Environmental Laboratories
- Food control: Confirmatory screening for unauthorized use in animal-derived products, ensuring compliance with EU zero-tolerance policies and national regulations.
- Environmental surveillance: Routine monitoring in wastewater, surface waters, and drinking water supply chains; assessment of treatment efficacy and risk management.
Why HPC Standards GmbH
- Specialist manufacturer and distributor focused on pesticides, veterinary products, metabolites, and stable isotope-labelled derivatives.
- Reliable lead times, rigorous QC, and technical support for method development, validation, and troubleshooting in accredited laboratories.


