New user? / Forgot your password?
0 Item | 0,00 €
Pyrimethamine — Reference Materials for Residue Analysis
Pyrimethamine High-Purity Reference Materials for Confident Compliance
Achieve traceable calibration and rock-solid method validation with HPC Standards high-purity pyrimethamine reference materials. Trusted by food and environmental laboratories, our neat solids and certified ready-to-use solutions support LCMSMS workflows, matrix-matched calibration, and routine QC at low gkg and ngL levels. Each lot ships with a comprehensive CoA purity, concentration, uncertainty, traceability and meets international quality requirements. Optional stable isotope-labelled derivatives help control matrix effects and improve quantification. Rely on fast delivery, expert technical support, and proven performance to meet MRL-driven demands with confidence.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
D3-Pyrimethamine solution | 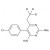 | 692455 | 1X1ML | |
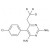 | 692454 | 1X5MG | ||
ISO 17034 Reference Material | 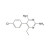 | 677479 | 1X250MG | Please log in. |
HPC Standards GmbH provides high-purity reference materials for pyrimethamine to support accurate calibration, method validation, and routine QC in food and environmental residue analysis. Our products are tested according to international quality requirements and meet the highest industrial standards.
Overview
Pyrimethamine (CAS 58-14-0) is a diaminopyrimidine antifolate that inhibits protozoan dihydrofolate reductase (DHFR). IUPAC: 5-(4-chlorophenyl)-6-ethyl-2,4-pyrimidinediamine. Molecular formula: C12H13ClN4; relative molecular mass ~248.7 g/mol.
Physicochemical profile: weak base (conjugate acid pKa ~7), low water solubility, higher solubility in acidic media and polar organics; low volatility; moderate lipophilicity. These properties inform extraction, cleanup, and detection strategies in residue analysis.
Uses and Applications
Human medicine: antimalarial and antiprotozoal (often combined with sulfonamides). Veterinary medicine: used against coccidial infections in companion animals and, historically, in some food-producing species under regulated conditions.
Relevance for residue testing: potential residues in animal-derived foods (e.g., meat, liver, kidney, milk, eggs) and occurrence in environmental waters near wastewater discharges or agricultural sites.
Regulatory Landscape
Pyrimethamine is subject to maximum residue limits (MRLs) in food of animal origin across many jurisdictions (e.g., EU, Codex, national frameworks). Compliance requires validated methods with fit-for-purpose LOQs aligned to applicable MRLs.
Laboratories should confirm current regional MRLs, target matrices, and marker residues, and document method performance (selectivity, trueness, precision, LOQ) per ISO/IEC and regulatory guidance.
Monitoring and Occurrence
Typical monitoring programs include targeted screening of animal products, feed, and wastewater/receiving waters. Elevated findings may occur near veterinary usage or from improper disposal of pharmaceuticals.
Trend analysis benefits from robust calibration with traceable reference materials and inclusion of internal standards to control matrix effects.
Analytical Methods
Preferred technique: LC–MS/MS in ESI positive mode, using multiple reaction monitoring for high sensitivity and selectivity. HPLC–UV/FLD may be used for certain clean matrices but typically with higher LOQs.
Method optimization: adjust mobile-phase pH (acidic) to improve peak shape; consider ion-pair-free systems compatible with MS; evaluate matrix effects and recovery via matrix-matched calibration and stable internal standards when available.
Sample Preparation
Food matrices: QuEChERS-based extraction with buffered salts, followed by dispersive SPE cleanup (e.g., PSA/C18) or cartridge SPE as needed for high-fat or high-protein matrices.
Environmental waters: SPE (e.g., HLB or mixed-mode sorbents) with acidified loading to enhance retention; elution with polar organics and subsequent concentration under controlled conditions.
Quality Control and Validation
Implement full validation (specificity, linearity, accuracy, precision, matrix effects, carryover, robustness, stability) per SANTE/ISO guidance. Include ongoing QC: system suitability, calibration verification, blanks, spikes, duplicates, and control charts.
Use certified reference materials to establish calibration traceability and evaluate recovery and bias across batches and matrices.
Health Impact — Human Toxicity
Mode of action: antifolate DHFR inhibition. Adverse effects at therapeutic or excessive exposure may include megaloblastic anemia, leukopenia, thrombocytopenia, and gastrointestinal or neurologic symptoms; folate deficiency increases risk. Pregnancy risk warrants careful assessment; leucovorin (folinic acid) is used clinically to mitigate antifolate toxicity.
Laboratory handling should minimize potential exposure via dermal contact, ingestion, or inhalation of dust.
Occupational Exposure and Safety Measures
Handle pyrimethamine as a potentially harmful pharmaceutical compound. Use lab coat, nitrile gloves, eye protection, and work in a fume hood or ventilated enclosure. Avoid aerosol/dust generation.
Storage: keep tightly closed in amber containers, dry, protected from light, at controlled temperature (typically 2–8 °C for solutions unless otherwise specified on the CoA). Dispose according to pharmaceutical/chemical waste regulations.
Environmental Impact
Fate: low volatility; potential persistence in aqueous environments depending on pH and light; adsorption to solids varies with organic content. Degradation may be slower in dark, neutral conditions.
Toxicity: antifolate activity can affect non-target microorganisms and primary producers; acute toxicity to aquatic invertebrates and algae is of concern at elevated levels. Bioaccumulation potential is low to moderate.
Effects on Wildlife and Ecology
Risk to aquatic communities includes disruption of microbial and algal growth, potentially altering trophic dynamics. Good wastewater management and responsible veterinary use help limit environmental loading.
Storage, Stability, and Handling of Standards
Neat material: stable if stored dry, cool, and protected from light. Avoid repeated freeze–thaw of solutions; aliquot upon receipt. Follow CoA/labels for exact storage temperature and shelf-life.
Solution stability depends on solvent, concentration, and container. Use amber glass and document in-use stability for calibration and QC solutions.
Matrices and Typical LOQs
Common matrices: muscle, liver, kidney, milk, eggs, feed, surface water, effluent, sediments, biota. Fit-for-purpose LOQs are typically in the low µg/kg (ppb) range for foods and ng/L–low µg/L for waters when using LC–MS/MS with SPE/QuEChERS cleanup.
Analytical Reference Materials
HPC Standards offers pyrimethamine reference materials as neat solids and ready-to-use certified solutions at various concentrations. Stable isotope-labelled derivatives may be available to support internal standardization and accurate quantification.
Each lot is accompanied by a Certificate of Analysis detailing purity, concentration (for solutions), uncertainty, traceability, and recommended storage. Products are tested according to international quality requirements and meet the highest industrial standards.
Compliance and Documentation
Our reference materials support ISO/IEC 17025 workflows, method validation, routine QC, and proficiency testing. Documentation includes CoA, safety data, and shipping records to facilitate audits and regulatory inspections.
Ordering and Logistics
Available packaging sizes accommodate screening through high-throughput monitoring. Shipped with temperature protection as required. Technical support is available for method development, matrix challenges, and selection of appropriate reference materials.


