New user? / Forgot your password?
0 Item | 0,00 €
Diaveridine Reference Materials for Residue Analysis
Diaveridine Precision Reference Materials for Confident Residue Compliance
Ensure defensible results at ppb levels with high-purity Diaveridine reference materials from HPC Standards. Manufactured and tested to international quality requirements, our neat and ready-to-use solutions support method development, matrix-matched calibration, QC, and proficiency testing in food and environmental laboratories. Optional isotopelabelled derivatives and metabolite standards enhance accuracy, mitigate matrix effects, and strengthen traceability for ISOIEC 17025 and ISO 17034 workflows. Benefit from lot-specific CoAs, temperature-controlled logistics, and expert technical guidanceso you can validate faster, monitor reliably, and meet regulatory limits with confidence.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
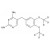 | 693050 | 1X10MG | Please log in. | |
D6-Diaveridine solution | 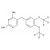 | 693051 | 1X1ML | |
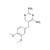 | 674885 | 1X100MG | Please log in. |
High-precision reference materials for Diaveridine to support compliant, defensible residue testing in food and environmental matrices.
Overview
Diaveridine is a diaminopyrimidine antimicrobial that inhibits dihydrofolate reductase. It is used in veterinary medicine, often in combination with sulfonamides, to enhance antibacterial and anticoccidial efficacy. In residue analysis, Diaveridine is monitored in edible tissues and products from treated animals to ensure compliance with regulatory limits.
As a target analyte, Diaveridine requires sensitive, selective quantification at low µg/kg (ppb) levels across complex matrices. Robust reference materials are essential for method development, calibration, quality control, and proficiency testing.
Chemical Class and Mechanism
Diaveridine belongs to the diaminopyrimidine class (DHFR inhibitors). It blocks tetrahydrofolate formation in microbes and protozoa, leading to impaired nucleic acid synthesis. In residue analytics, the basic diaminopyrimidine core influences extraction, ionization efficiency, and chromatographic behavior.
Analytical implications include pH-dependent partitioning, potential ionic interactions with sorbents, and predictable MS/MS fragmentation pathways typical for diaminopyrimidines.
Uses
Veterinary applications include combination therapies with sulfonamides for the prevention and treatment of bacterial infections and coccidiosis in food-producing animals. These uses can result in residues in muscle, liver, kidney, fat, milk, and eggs if withdrawal periods are not respected.
Analytical laboratories monitor Diaveridine to verify adherence to good veterinary practice, withdrawal intervals, and legal residue limits.
Regulatory Status and MRLs
Regulatory frameworks (e.g., EU, Codex, national authorities) may establish maximum residue limits (MRLs) or tolerance levels for Diaveridine in specified animal-derived matrices. Status and MRLs can vary by jurisdiction and commodity.
Laboratories should consult current legislation, databases, and veterinary product authorizations to confirm applicable MRLs, target analytes (parent and metabolites), and decision limits.
Residue Monitoring and Target Matrices
Typical target matrices include muscle, liver, kidney, fat/skin, milk, and eggs. Surveillance programs may also assess feed and environmental samples in certain contexts.
Screening and confirmatory workflows should be designed to achieve limits of quantification (LOQs) at or below relevant MRLs, with appropriate matrix extensions validated where required.
Sample Preparation and Extraction
Common approaches include protein precipitation and solid–liquid extraction using aqueous organic solvents, with pH control to optimize recovery of basic analytes. Clean-up may involve dispersive SPE (e.g., QuEChERS variants with PSA/C18) or cartridge SPE to reduce matrix effects.
Method optimization focuses on recovery, suppression/ion enhancement control, and ruggedness across diverse matrices. Stable, traceable reference materials are critical for extraction recovery checks and day-to-day performance tracking.
Analytical Methods
LC–MS/MS is the preferred confirmatory technique due to selectivity and sensitivity, employing reversed-phase chromatography and multiple reaction monitoring (MRM). HPLC–UV/FLD may be used for screening where sensitivity allows.
Key parameters include: retention stability, two or more diagnostic transitions, isotopically labelled internal standards for matrix effect compensation, and calibration in matrix-matched or standard-addition formats.
Reference Materials from HPC Standards
HPC Standards provides high-purity Diaveridine reference materials for calibration, quality control, and method validation in food and environmental analysis. Products are supplied with comprehensive Certificates of Analysis, including purity, identity, uncertainty, and traceability information according to international quality requirements.
Available formats include neat materials and ready-to-use solutions at defined concentrations. Upon request, we offer custom mixtures and matrix spikes to support multi-residue workflows.
Isotope-Labelled and Metabolite Options
To improve quantitative accuracy and correct for matrix effects, HPC Standards offers stable isotope-labelled derivatives where available. Metabolite and related-compound reference materials can be supplied to broaden scope and verify potential co-eluting or transformant species.
Including labelled internal standards in every analytical batch enhances precision, trueness, and measurement uncertainty control.
Health Impact (Human Toxicity)
As a DHFR inhibitor, Diaveridine can affect folate pathways at sufficient exposure. Laboratory handling should minimize ingestion, inhalation, or dermal exposure. In residue terms, consumer risk assessments underpin MRL settings and withdrawal periods to ensure dietary safety.
Follow institutional chemical hygiene policies and consult the Safety Data Sheet (SDS) for hazard statements and first-aid guidance.
Environmental Impact and Fate
Veterinary actives may reach the environment via excreta and manure application. Potential effects include antimicrobial pressure on microbial communities and risks to non-target organisms depending on concentrations and persistence.
Environmental monitoring, when applicable, should assess relevant compartments (soil, water, sediment) with validated LC–MS/MS methods and suitable reference materials for calibration and quality control.
Effects on Wildlife
Exposure of non-target species can occur indirectly through contaminated media. Risk characterization depends on local usage patterns, degradation, and bioavailability. Data-driven surveillance supports targeted mitigation where necessary.
Safety Measures (Laboratory)
Handle reference materials in a fume hood with appropriate PPE (lab coat, nitrile gloves, safety glasses). Avoid dust and aerosols. Use calibrated pipettes for solutions to prevent dosing errors. Dispose of waste according to institutional and regulatory requirements.
Implement spill control procedures, label all containers clearly, and segregate incompatible chemicals per SDS guidance.
Quality Assurance and Compliance
HPC Standards products support ISO/IEC 17025 and ISO 17034 compliant workflows. Use certified reference materials for calibration, control charts (QC), and proficiency testing participation to demonstrate method performance and traceability.
Document recovery, repeatability, reproducibility, measurement uncertainty, and decision rules in line with accreditation requirements.
Method Validation and Performance
Validate selectivity, linearity, recovery, matrix effects, LOQ/LQ, precision, trueness, and stability. Employ matrix-matched calibration with isotope-labelled internal standards where available. Conduct robustness tests across instruments, analysts, and days.
Ongoing verification with independent QC levels and proficiency tests ensures sustained compliance and defensible reporting.
Stability, Storage, and Handling of Reference Materials
Store Diaveridine reference materials in tightly closed, inert containers, protected from light, typically refrigerated or frozen as specified on the CoA/SDS. Allow solutions to equilibrate to room temperature before opening to prevent condensation.
Track aliquot history, minimize freeze–thaw cycles, and adhere to assigned shelf life and uncertainty statements.
Reporting and Uncertainty
Report results with associated measurement uncertainty and decision rules relative to MRLs or action limits. Maintain traceability from calibration standards to final reported values, including batch identifiers and CoA references.
Use appropriate rounding and significant figures consistent with method capability and regulatory guidance.
Packaging, Logistics, and Documentation
HPC Standards supplies Diaveridine reference materials in amber vials with tamper-evident seals. Temperature-controlled shipping and lot-specific documentation (CoA, SDS) ensure integrity upon arrival.
Custom concentrations, multi-analyte mixes, and bulk quantities are available to align with laboratory throughput and method needs.
Ordering Information
Contact our technical team for product selection, isotope-labelled options, and method support. We provide guidance on matrix selection, calibration strategy, and QC design to accelerate validation and routine implementation.
HPC Standards is your partner for reliable reference materials covering veterinary drugs, pesticides, metabolites, and isotope-labelled derivatives to meet the highest industrial and regulatory standards.


