New user? / Forgot your password?
0 Item | 0,00 €
Citalopram hydrobromide
Citalopram hydrobromide High-Purity Reference Materials for Confident LCMS Quantification
Achieve trace-level accuracy in pharmaceutical QC and environmental monitoring with HPC Standards citalopram hydrobromide reference materials. Produced to international quality requirements, our neat and ready-to-use solutions deliver reliable calibration, method validation, and proficiency testing across water, sludge, and product matrices. Complement with stable isotopelabelled analogues and related analytes e.g., escitalopram, N-desmethylcitalopram for robust isotopedilution workflows. Count on documented purity, uncertainty, and tailored concentrations to meet your labs compliance and performance targets.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 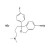 | 687742 | 1X50MG | Please log in. |
ISO 17034 Certified Reference Material Citalopram hydrobromide solution | 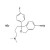 | 688599 | 1X5ML | Please log in. |
Precision reference materials for confident quantification of citalopram hydrobromide in pharmaceutical and environmental matrices.
Overview
Citalopram hydrobromide is the hydrobromide salt of the selective serotonin reuptake inhibitor (SSRI) citalopram, widely used as an antidepressant. In analytical contexts, it serves as a target compound for pharmaceutical quality control and for monitoring pharmaceutical residues in wastewater, surface water, and sludge.
High-quality reference materials are essential for accurate calibration, method validation, proficiency testing, and routine surveillance of citalopram and related compounds at trace levels.
Chemical Identity and Properties
Chemical name: Citalopram hydrobromide (salt of citalopram). Common synonyms: 1-(3-dimethylaminopropyl)-1-(4-fluorophenyl)-1,3-dihydroisobenzofuran-5-carbonitrile hydrobromide.
Molecular formula (salt): C20H22BrFN2O. Approximate molecular weight: ~405.3 g/mol (salt). The hydrobromide salt is water-soluble and suitable for aqueous and polar organic solvent systems; the free base is more lipophilic.
Structural features include a fluorophenyl group and a dihydroisobenzofuran scaffold; the marketed active enantiomer is S-citalopram (escitalopram), relevant for chiral analyses.
Uses and Occurrence
Primary use: active pharmaceutical ingredient (API) in human medicine as an SSRI. It is frequently detected in municipal wastewater effluents and receiving waters due to incomplete removal in conventional treatment.
Analytical interest spans pharmaceutical manufacturing QC, stability studies, and environmental monitoring programs assessing pharmaceutical residues in influents, effluents, surface waters, sediments, and sludge.
Regulatory and Guidance Context
Pharmaceutical residue monitoring is driven by environmental quality initiatives, permitting requirements, and risk assessments. While citalopram is not a global priority pollutant, it is included in many regional watch lists and investigative monitoring programs for pharmaceuticals in water.
Analytical laboratories support compliance with international quality requirements and customer-specific specifications for APIs and for wastewater/surface-water monitoring programs, following validated methods and traceability to certified reference materials where applicable.
Monitoring and Analytical Methods
Quantification is commonly performed by LC–MS/MS or LC–HRMS for selectivity and sensitivity; HPLC–UV may be used for higher concentration ranges in QC. Chiral LC can resolve citalopram enantiomers and differentiate from escitalopram where required.
Typical workflows employ solid-phase extraction (SPE) for water matrices, followed by LC–MS/MS with isotope-dilution using stable isotope-labelled internal standards to correct for matrix effects and recovery.
Sample Preparation and Matrices
Water: SPE (e.g., reversed-phase or mixed-mode) with eluates concentrated prior to LC–MS/MS. Sludge/sediment: solvent extraction (e.g., methanol or acetonitrile-based), cleanup, and concentration. Pharmaceutical products: dissolution/extraction in suitable solvents and filtration before analysis.
Stability during sampling and storage is enhanced by cooling, protection from light, and use of amber containers; pH control may be applied per validated methods.
Quality and Traceability
Accurate quantification requires traceable reference materials with known purity and uncertainty. Use of matrix-matched calibrations and stable isotope-labelled internal standards supports measurement accuracy, precision, and method robustness.
HPC Standards reference materials are produced and tested according to international quality requirements and meet high industrial standards to support method validation, system suitability testing, and proficiency assessments.
Health Impact (Human Toxicity)
Citalopram is a centrally acting SSRI. Toxicological concerns at excessive exposure include serotonergic effects (e.g., serotonin syndrome), gastrointestinal symptoms, CNS effects, and potential QT interval prolongation at high doses. Occupational exposure in laboratories should be minimized.
Implement controls to prevent ingestion, inhalation of dust/aerosols, and skin/eye contact. Follow institutional safety protocols and consult the material’s safety data sheet for detailed hazard information specific to your lot/formulation.
Environmental Impact
Citalopram is detected in aquatic environments at ng/L to low µg/L levels near urban discharges. It exhibits moderate polarity (as hydrobromide salt) and can persist through conventional treatment to varying degrees, depending on process conditions.
Degradation and sorption behavior are matrix- and pH-dependent; transformation products (e.g., N-desmethylcitalopram) may form and should be considered in comprehensive monitoring programs.
Effects on Wildlife
SSRIs can interact with serotonin signaling in non-target aquatic organisms. Reported sublethal effects for related compounds include behavioral and physiological changes in fish and invertebrates. Site-specific risk assessments should consider local concentrations, species sensitivity, and mixture effects.
Safety and Handling
Handle reference materials in a fume hood with appropriate PPE (lab coat, nitrile gloves, safety glasses). Avoid dust generation and aerosol formation. Use calibrated equipment and validated procedures to prevent cross-contamination and ensure data integrity.
Dispose of waste according to institutional guidelines and local regulations for pharmaceutical substances and organic solvents.
Storage and Stability
Store neat materials tightly closed in a cool, dry, and well-ventilated area, protected from light. Solution reference materials are typically stored refrigerated or frozen according to the certificate or label to preserve stability.
Allow solutions to equilibrate to room temperature before opening to prevent moisture ingress and concentration shifts. Observe stated shelf lives and requalification dates.
Reference Materials from HPC Standards
HPC Standards provides high-purity reference materials for citalopram hydrobromide to support accurate and reliable residue analysis in pharmaceutical QC and environmental monitoring.
Available formats typically include neat materials and ready-to-use solutions in common solvents at application-relevant concentrations. Stable isotope-labelled internal standards and related analytes are available to enhance analytical accuracy.
Related Analytes and Isotopologues
Complementary reference materials include citalopram (free base), escitalopram (S-enantiomer), N-desmethylcitalopram, and other transformation products, as well as deuterated or 13C/15N-labelled analogues for isotope-dilution workflows.
Chiral reference materials can support enantioselective methods where differentiation between citalopram and escitalopram is required.
Applications in QA/QC and Method Validation
Use in calibration curves, system suitability tests, spike-recovery studies, matrix effects evaluation, limit of detection/quantification determination, and interlaboratory comparisons.
Supports compliance with internal specifications, client requirements, and international quality frameworks for analytical measurement.
Key Identifiers
CAS (citalopram hydrobromide): 59729-32-7. CAS (citalopram free base): 59729-33-8. Selected synonyms and identifiers are provided on product documentation to ensure unambiguous selection and traceability.
Ordering Information
Products are supplied with detailed documentation including purity, concentration (for solutions), uncertainty (where applicable), safety information, and recommended storage conditions. Custom concentrations, solvent systems, and multi-analyte mixes are available on request.


