New user? / Forgot your password?
0 Item | 0,00 €
Triflumezopyrim Reference Materials for Residue Analysis
Triflumezopyrim Reference Materials Precision for Regulatory-Ready Residue Analysis
Achieve confident, reproducible quantification with high-purity Triflumezopyrim reference materials from HPC Standards. Engineered for LC-MSMS workflows, our neat solids, ready-to-use solutions, and optional isotope-labelled analogs streamline calibration, spike preparation, and method validation across food and environmental matrices. Each lot is produced to international quality requirements and delivered with a comprehensive Certificate of Analysis covering purity, identity, concentration, uncertainty, and traceabilitysupporting accreditation, MRL compliance, and robust QC. Custom concentrations, solvent systems, and multi-analyte mixes available to optimize your laboratory throughput.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 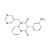 | 684916 | 1X10MG | Please log in. |
ISO 17034 Certified Reference Material Triflumezopyrim solution | 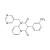 | 685710 | 1X1ML | Please log in. |
High-purity reference materials for Triflumezopyrim to support precise quantification in food and environmental testing, method validation, and regulatory compliance.
Overview
Triflumezopyrim is a mesoionic insecticide belonging to the nicotinic acetylcholine receptor (nAChR) modulator class. It is designed for effective control of planthoppers and related sap-feeding pests, particularly in rice production systems. Its distinct chemistry enables rapid feeding cessation in target insects and supports resistance management strategies when used within integrated pest management programs.
Chemical Class and Mode of Action
Class: nAChR modulators (IRAC Group 4; mesoionic subgroup). Triflumezopyrim acts on insect nAChRs, disrupting synaptic transmission and leading to quick feeding inhibition and mortality in susceptible species. Its binding profile differs from neonicotinoids, offering utility where cross-resistance management is required.
Uses and Target Pests
Primary targets include brown planthopper (Nilaparvata lugens) and white-backed planthopper (Sogatella furcifera) in rice. Additional activity has been reported against other hemipteran pests in various crops, depending on local registrations. It is positioned for foliar or seedling-stage protection in high-pressure rice ecosystems and can be integrated with other control measures to mitigate resistance development.
Regulatory Status and MRLs
Registration status varies by country and crop. Maximum Residue Limits (MRLs) are established in several jurisdictions for matrices such as rice, vegetables, and fruits. Laboratories should consult the latest national and international databases (e.g., Codex, US EPA tolerance tables, Japan MHLW, EU pesticide database) for current MRLs and approved uses. Note that approval status may differ across markets; verify local regulatory requirements prior to monitoring and reporting.
Human Health and Toxicity
Available regulatory summaries indicate low to moderate acute toxicity in mammals with typical handling precautions required for pesticides. Potential for eye and skin irritation exists; avoid inhalation and ingestion. Always adhere to institutional risk assessments and consult the Safety Data Sheet for detailed toxicological endpoints, first-aid measures, and exposure controls.
Environmental Impact
Triflumezopyrim exhibits high activity toward target hemipterans and may pose hazards to aquatic invertebrates; implement drift and runoff mitigation. Reported selectivity toward beneficial organisms varies with exposure route and formulation. Follow label constraints and local environmental protection guidelines to minimize non-target exposure and preserve beneficial arthropods and pollinators.
Residue Monitoring in Food and Environment
Common matrices: rice grain, husk, straw, paddy water, irrigation water, vegetables, fruits, soil, and sediment. Monitoring focuses on parent compound quantification; some programs may include selected transformation products based on jurisdictional requirements. Typical LOQs for food matrices range from low to sub-ppb depending on instrumentation and cleanup strategy.
Analytical Methods
LC-MS/MS is the method of choice for specificity and sensitivity. Sample preparation frequently employs QuEChERS-based extraction with matrix-appropriate cleanup (e.g., PSA, C18, GCB as needed). Isocratic or gradient reversed-phase separation with electrospray ionization is common. Method validation should establish specificity, linearity, matrix-matched calibration, recovery (spike levels across MRL-relevant ranges), repeatability, intermediate precision, and measurement uncertainty.
HPC Standards Reference Materials for Triflumezopyrim
HPC Standards GmbH supplies high-purity reference materials for Triflumezopyrim to support quantitative residue analysis in food and environmental laboratories. Materials are produced and tested according to international quality requirements and are accompanied by a detailed Certificate of Analysis specifying purity, identity confirmation, concentration (for solutions), uncertainty, and traceability.
Available formats include neat solids and ready-to-use solutions in suitable solvents to facilitate calibration and spike preparation. Stable isotope-labelled analogs and selected related compounds may be available on request to enhance accuracy in complex matrices through isotope dilution or as internal standards.
Quality and Documentation
Each lot is characterized using orthogonal techniques (e.g., LC-UV, LC-MS, NMR where applicable) to confirm identity and purity. CoAs include test methods, chromatographic purity, concentration verification for solutions, storage conditions, and shelf-life information. Batch-specific documentation supports method validation, routine QC, and accreditation needs.
Storage and Stability
Store reference materials in original containers, tightly closed, protected from light and moisture, and under recommended temperature conditions as stated on the label and Certificate of Analysis. Allow solutions to equilibrate to room temperature before opening to minimize condensation. Observe expiry dates and manage stock under documented inventory control for traceable usage.
Safety and Handling
Handle Triflumezopyrim reference materials in a chemical fume hood with appropriate PPE: lab coat, safety glasses, and suitable gloves. Avoid skin and eye contact and prevent inhalation of vapors or aerosols. Dispose of waste according to institutional and regulatory requirements for pesticide-containing materials.
Method Development and Validation Support
Our reference materials facilitate calibration curve preparation, recovery studies, determination of matrix effects, and verification of LOQs/MDLs. Matrix-matched calibration and use of internal or isotope-labelled standards are recommended to mitigate ion suppression or enhancement in LC-MS/MS.
Compliance and Reporting
Align analytical scope with regional MRLs and action limits. Apply appropriate quality controls (procedural blanks, fortified blanks, matrix spikes, duplicate samples, and control charts). Report results with associated measurement uncertainty, identification criteria, and confirmation data where required by regulation.
Applications by Matrix
Food: rice, leafy and fruiting vegetables, fruits, processed products. Environment: surface and irrigation water, soil, sediment, and paddy-field compartments. Occupational and product QC: formulation checks and contamination control during manufacturing or handling.
Related Compounds and Metabolites
Depending on regulatory scope and risk assessment, laboratories may extend monitoring to selected transformation products. Contact HPC Standards for availability of related reference materials to support comprehensive residue profiling and method robustness.
Ordering, Packaging, and Shipping
Reference materials are supplied in tamper-evident packaging with clear labeling, storage guidance, and batch traceability. Temperature-controlled shipping is available where required. Custom concentrations, solvent systems, and multi-analyte mixes can be prepared on request to streamline laboratory workflows.
Why HPC Standards
We are a dedicated manufacturer and distributor of reference materials for pesticides, veterinary drugs, their metabolites, and stable isotope-labelled derivatives. Our products meet stringent industrial standards and support laboratories in achieving reliable, reproducible results and regulatory compliance in food and environmental analysis.


