New user? / Forgot your password?
0 Item | 0,00 €
Bisphenol B (BPB) — Reference Materials for Residue and Migration Analysis
Bisphenol B Precision Reference Materials for Confident Compliance
Ensure defensible results with HPC Standards highpurity Bisphenol B BPB reference materials for residue and migration analysis. Our traceable neat solids and certified readytouse solutions support method development, validation, and routine monitoring across foodcontact, environmental, and industrial matrices. Delivered with batchspecific CoA, documented purity and uncertainty, and options for custom mixes and isotopelabelled standards, we help accredited labs meet EU FCM, REACH, and NIAS expectationson time, every time.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
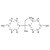 | 693721 | 1X5MG | ||
13C12-Bisphenol B solution | 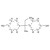 | 693722 | 1X1ML | |
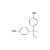 | 676798 | 1X100MG | Please log in. | |
Bisphenol B solution | 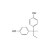 | 685557 | 1X1ML | Please log in. |
High-quality reference materials for precise identification, calibration, and quantification of Bisphenol B in food-contact, environmental, and industrial matrices. Manufactured and tested to international quality requirements.
Overview
Bisphenol B (BPB; 2,2-bis(4-hydroxyphenyl)butane) is a bisphenol analogue used as a monomer or additive in polymeric materials and industrial applications. It can occur as a residue, impurity, or degradation product in plastics, coatings, and resins and may migrate into food or the environment.
HPC Standards provides traceable reference materials for BPB to support robust method development, validation, and routine monitoring in accredited laboratories.
Chemical Identity & Properties
CAS: 77-40-7; Synonyms: 2,2-bis(p-hydroxyphenyl)butane; Molecular formula: C16H18O2; Approx. molar mass: 242.31 g/mol.
Physicochemical profile (typical): weakly acidic phenolic compound; low water solubility; moderate hydrophobicity. Relevant for partitioning into organic phases and sorption to particulates and polymers.
Uses & Sources
Potential uses include monomer or modifying agent in epoxy/polycarbonate-type systems, resin formulations, coatings, and thermal paper alternatives. BPB may be present as a manufacturing residual, processing aid, or contaminant in materials and articles.
Analytical interest arises from substitution of BPA with structural analogues, requiring comprehensive surveillance of bisphenol residues and migrants.
Regulatory Landscape
BPB is scrutinized as a BPA analogue in food-contact and environmental monitoring programs. Specific regulatory limits for BPB vary by jurisdiction and application; where not explicitly listed, risk assessments and customer-specific specifications often apply.
Compliance frameworks commonly referenced include EU food-contact materials legislation, REACH obligations, national chemical inventories, and sectoral guidance on non-intentionally added substances (NIAS). Laboratories should verify current local requirements before reporting.
Health Impact — Human Toxicity & Endocrine Activity
In vitro data indicate estrogenic activity for several bisphenol analogues, including BPB. Toxicological datasets for BPB are comparatively limited versus BPA; precautionary assessment typically considers potential endocrine-disrupting properties, with attention to sensitive populations.
Risk characterization relies on migration or exposure data combined with potency information. Robust quantification using validated reference materials is essential to support reliable exposure assessment.
Environmental Impact — Fate and Effects on Wildlife
BPB may enter aquatic systems via industrial effluents, landfill leachates, or product weathering. Expected behavior includes sorption to sediments/solids and occurrence in surface waters at low ng/L–µg/L levels when present.
Ecotoxicological data for BPB are emerging; as with other bisphenols, potential sublethal effects (endocrine-related endpoints) are of interest. Environmental monitoring in water, sediment, sludge, and biota is recommended when BPB is relevant to site activities.
Sampling & Sample Preparation
Typical matrices: polymer articles, food simulants (migration studies), drinking/surface/groundwater, wastewater/effluent, sludge/sediment, indoor dust, and biological samples.
Common approaches: solvent extraction or solid–liquid extraction for solids; SPE (e.g., polymeric reversed-phase) for waters; QuEChERS-type workflows for complex matrices; migration testing per standardized conditions and simulants. Use BPB reference materials to prepare matrix-matched calibrants and spikes.
Analytical Methods
LC–MS/MS with electrospray ionization is widely applied for bisphenols, enabling sensitive and selective quantification without derivatization. Key transitions target phenolic deprotonated ions with characteristic fragments.
GC–MS(/MS) may be employed after silylation or acylation derivatization to enhance volatility. Chromatographic baselines should minimize carryover and phenolic adsorption; utilize low-adsorption vials and tubing.
Reference Materials from HPC Standards
Available formats: neat BPB solid and ready-to-use certified solutions (e.g., 100 µg/mL or custom concentrations in acetonitrile or methanol). Custom mixes with BPA, BPF, BPS, and other analogues on request.
Each lot is manufactured under stringent quality control with full documentation (Certificate of Analysis, traceability, purity profile, uncertainty where applicable) to support ISO/IEC 17025 and ISO 17034 workflows.
Isotope-Labelled Reference Materials
Stable isotope-labelled derivatives for BPB (subject to availability) are recommended as internal standards to correct for matrix effects, recovery, and ion suppression. Please inquire for current availability, target isotopic enrichment, and solvent systems.
Calibration & Quality Control Strategy
- Multi-level calibration across anticipated concentration ranges (e.g., low ng/L to µg/L for waters; ng/g for solids).
- Use of isotopically labelled internal standards or structurally close surrogates when labelled BPB is unavailable.
- Routine QC: method blanks, solvent blanks, matrix spikes, duplicates, and continuing calibration verification.
Include recovery studies and measurement uncertainty estimation to align with accreditation and client reporting needs.
Matrices & Application Areas
- Food-contact materials and migration simulants (ethanol, acetic acid, oil simulants).
- Environmental waters, WWTP influent/effluent, sludge, and sediments.
- Indoor dust and wipes for product/material assessment.
- Product stewardship, NIAS screening, and supply-chain compliance.
Storage & Stability
Store BPB reference materials at 2–8 °C in amber glass, protected from light and moisture. Allow solutions to equilibrate to room temperature before opening. Adhere to expiration dates and re-test intervals specified on the certificate.
Minimize freeze–thaw cycles; prepare aliquots for routine analysis to preserve integrity.
Safety Measures & Handling
Handle BPB as a potentially endocrine-active phenolic compound. Use appropriate PPE (lab coat, safety glasses, nitrile gloves) and work in a fume hood. Avoid skin contact and inhalation of dust or aerosols.
Dispose of waste according to institutional and local regulations. Consult the Safety Data Sheet for hazard information and first-aid measures.
Documentation & Compliance Support
Delivered with batch-specific Certificate of Analysis and product documentation supporting method validation, traceability, and audit readiness. Upon request, we provide supplier declarations to assist with regulatory submissions.
Shipping, Packaging & Lead Times
Products are shipped in validated, leak-proof packaging with temperature control where required. Standard concentrations and neat materials are typically available with short lead times; custom mixtures and concentrations upon request.
Related Compounds and Mixes
Complementary reference materials: Bisphenol A (BPA), Bisphenol S (BPS), Bisphenol F (BPF), and additional analogues and metabolites. Ready-to-use multicomponent bisphenol mixes are available for screening and quantitative workflows.
Monitoring & Reporting Guidelines
Align analytical plans with current guidance for bisphenols in food-contact materials, environmental waters, and NIAS risk assessment. Report results with calibration model, recovery, LOQ/LOD, measurement uncertainty, and sample history to ensure regulatory defensibility.
Why HPC Standards
As a manufacturer and distributor of high-quality reference materials for pesticides, veterinary drugs, industrial chemicals, and isotope-labelled derivatives, HPC Standards is your competent partner for food and environmental analysis. Our products meet international quality requirements and the highest industrial standards, ensuring reliable results and regulatory compliance.


