New user? / Forgot your password?
0 Item | 0,00 €
Epitetracycline Hydrochloride — Reference Materials for Residue Analysis
Epitetracycline hydrochloride Precision Reference Materials for Confident Compliance
Drive accuracy in tetracycline residue monitoring with HPC Standards high-purity epitetracycline hydrochloride. Our traceable reference materialsavailable as neat substances or ready-to-use solutionsare manufactured and quality-controlled to international requirements, supporting ISOIEC 17025 workflows. Achieve robust LCMSMS calibration, verify epimer separation, and stabilize epimer ratios across food, feed, and environmental matrices. Each lot ships with a comprehensive COA, SDS, and stability guidance for audit-ready documentation, enabling reliable method validation, routine QC, and proficiency testing. Choose HPC Standards for proven expertise in veterinary drug epimers and uncompromising analytical performance.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 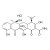 | 689977 | 1X10MG | Please log in. |
High-quality reference materials for epitetracycline hydrochloride designed for robust quantification and confirmation in food and environmental residue testing. Manufactured and quality-controlled to international standards for confident regulatory compliance.
Overview
Epitetracycline hydrochloride is the hydrochloride salt of 4-epitetracycline, an epimeric transformation product of tetracycline-class antibiotics. It commonly forms via pH- and temperature-dependent epimerization of tetracycline and may co-occur with parent compounds in food-producing animals, aquaculture, and environmental matrices.
As an epimer, epitetracycline typically exhibits reduced antimicrobial activity compared to tetracycline; however, it is analytically relevant due to regulatory frameworks that consider the sum of tetracyclines and their epimers for compliance monitoring.
Chemical Identity and Properties
- Class: Tetracycline antibiotic epimer (hydrochloride salt)
- Typical characteristics: amphoteric, light- and pH-sensitive; forms chelates with divalent/trivalent cations; moderate polarity suitable for reversed-phase LC separations
- Transformation: forms from tetracycline via epimerization; interconversion can be matrix- and storage-dependent
Uses in Analytical Context
- Calibration and system suitability standard for LC–MS/MS or LC–DAD methods targeting tetracycline antibiotics and their epimers
- Identification and quantitation of degradation/impurity profiles in pharmaceutical, veterinary, and feed matrices
- Spike-recovery experiments, method validation (LOD/LOQ, linearity, trueness, precision), and proficiency testing in food and environmental labs
Regulatory Context
- Many jurisdictions regulate the sum of tetracyclines (e.g., tetracycline, oxytetracycline, chlortetracycline) and their 4-epimers in edible tissues, milk, eggs, honey, and aquaculture products
- Reference frameworks include EU veterinary residue regulations and Codex guidance; national programs frequently mandate monitoring of epimers alongside parent drugs
- Laboratories require validated methods and traceable reference materials to demonstrate conformity with maximum residue limits (MRLs) and to support official control programs
Monitoring in Food and Environment
- Food: muscle, liver, kidney, milk/dairy, eggs, honey, fish/shrimp and other aquaculture products
- Environmental: surface water, sediments, wastewater/effluents, manure, sludge-amended soils
- Drivers: antimicrobial stewardship, surveillance of veterinary drug use, and risk management of resistance development
Human Toxicity
- Tetracycline-class effects may include gastrointestinal disturbance, photosensitivity, and hypersensitivity reactions; class-related concerns for teeth/bone development in children
- Epitetracycline exhibits lower antimicrobial potency than tetracycline but remains relevant for cumulative exposure assessments
- Occupational exposure in labs should be minimized via engineering controls and PPE
Environmental Impact
- Fate: adsorption to soils/sediments, potential persistence under certain conditions, photolysis and biotransformation possible
- Effects: alteration of microbial communities, selection pressure for antimicrobial resistance; toxicity reported for algae and aquatic microorganisms at sufficient concentrations
- Monitoring supports risk assessments in water bodies affected by agricultural and aquaculture discharges
Degradation and Transformation
- Epimerization (tetracycline ⇌ epitetracycline) is pH- and temperature-dependent; can occur during sample storage and preparation
- Additional pathways: dehydration, oxidation, complexation with metal ions; these can impact analytical recoveries and signal response
Stability and Handling
- Store reference materials in tightly closed, light-protective containers at low temperature as specified on the certificate of analysis (e.g., 2–8 °C or ≤−18 °C)
- Avoid alkaline conditions and prolonged exposure to elevated temperatures; use metal-free solvents or control for chelating effects
- Prepare fresh working solutions; document freeze–thaw cycles and solution stability as part of method SOPs
Safety Measures and Storage
- Use appropriate PPE (lab coat, gloves, eye protection) and handle in a fume hood or well-ventilated area
- Avoid inhalation, ingestion, and skin contact; follow local regulations for antibiotic waste disposal
- Refer to the product safety data sheet (SDS) for hazard statements and first-aid measures
Analytical Methods
- LC–MS/MS (ESI+): preferred for selectivity and sensitivity; multiple reaction monitoring of epimer-specific transitions
- LC–UV/DAD: applicable for higher-level screening; less selective than MS-based detection
- Sample preparation: protein precipitation for milk; SPE (HLB/cation-exchange) or modified QuEChERS for tissues, honey, and aquatic matrices; include chelator or buffer to stabilize epimer ratios
- Quality controls: matrix-matched calibration, isotopically labeled internal standards where available, recovery checks and bracketing standards
Reference Materials from HPC Standards
- Epitetracycline hydrochloride reference materials for quantitative residue analysis across food and environmental matrices
- Options: neat material or solution formats; accompanied by certificate of analysis with purity, identity, and uncertainty information
- Manufactured and tested according to international quality requirements to meet the highest industrial standards; suitable for method validation, routine QC, and proficiency testing support
Matrix Applications
- Food: bovine/porcine/poultry muscle, liver, kidney, milk, eggs, honey, fish/shrimp
- Environmental: surface/ground water, effluents, sediments, soils, biosolids
- Feed and premixes: impurity/degradation profiling during stability studies
Quality and Compliance Support
- Batch traceability, documented purity and identity testing, and stability guidance enable audit-ready compliance
- Technical documentation supports accreditation to ISO/IEC 17025 methods and aligns with regulatory residue monitoring plans
Method Development Tips
- Control pH during extraction and analysis to minimize epimer interconversion
- Evaluate matrix effects and ion suppression; use matrix-matched calibration and internal standards when possible
- Verify epimer separation and response factors; include system suitability criteria for retention time and ion ratios
Logistics and Availability
- Secure packaging in amber vials to protect from light; temperature-controlled shipping available
- Lot-specific COA, SDS, and technical notes provided; global delivery to accredited laboratories and industry
Why HPC Standards
- Expertise in reference materials for veterinary drugs, pesticides, metabolites, and stable isotope-labelled derivatives
- Dedicated support for food and environmental analysis with products designed to ensure regulatory compliance and analytical reliability


