New user? / Forgot your password?
0 Item | 0,00 €
Praziquantel Reference Materials
Praziquantel High-Purity Reference Materials for Confident Residue Analysis
Accelerate compliant, traceable results with Praziquantel reference materials from HPC Standards. Our high-precision neat substances and ready-to-use solutions including key metabolites and stable isotopelabelled derivatives enable accurate identification, quantification, and method validation across challenging matrices such as aquaculture products, livestock tissues, feed, and environmental samples. Produced under stringent international quality requirements and delivered with comprehensive CoAs, these materials support LCMSMS workflows, chiralseparation needs, matrixmatched calibration, and ISOSANTEaligned validation. Rely on HPC Standards for robust calibration, lowlevel detection, and custom concentrations or mixes tailored to your multi-residue methods.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
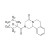 | 679428 | 1X10MG | Please log in. | |
D11-Praziquantel solution | 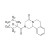 | 690862 | 1X1ML | Please log in. |
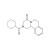 | 676882 | 1X250MG | Please log in. |
High-precision reference materials for Praziquantel and key metabolites to support reliable residue testing in food and environmental matrices.
Overview
Praziquantel is a broad-spectrum anthelmintic active against trematodes (flukes) and cestodes (tapeworms). It is widely used in human medicine for schistosomiasis and other trematode/cestode infections, and in veterinary medicine including aquaculture. Chemical name: 2-(Cyclohexanecarbonyl)-1,2,3,6,7,11b-hexahydro-4H-pyrazino[2,1-a]isoquinolin-4-one; CAS: 55268-74-1; molecular formula: C19H24N2O2. Commercial drug material is typically a racemate; the (R)-enantiomer is primarily responsible for activity.
HPC Standards provides Praziquantel reference materials designed for accurate identification, quantification, and method validation in residue analysis.
Uses
Human health: first-line therapy for schistosomiasis; also used for clonorchiasis, opisthorchiasis, paragonimiasis, and taeniasis. Veterinary: control of tapeworms and certain flukes in companion animals and food-producing species; frequent application in aquaculture for fish helminths.
Due to its veterinary use and potential residues in edible tissues, Praziquantel is a routine target in food safety monitoring programs.
Regulatory and MRLs
Maximum residue limits (MRLs) for Praziquantel in food-producing species are established in several jurisdictions (e.g., EU, Codex, and national frameworks). In the EU, listings under the veterinary drug residue legislation specify permitted uses, target species, and residue definitions for monitoring. Laboratories should consult current, jurisdiction-specific regulations for applicable MRLs and residue definitions (parent compound and/or specified metabolites).
Regulatory compliance requires validated methods with appropriate selectivity, sensitivity, and traceability, supported by fit-for-purpose reference materials.
Monitoring and Target Matrices
Typical matrices: fish muscle/skin, fish liver, shrimp and other aquaculture products; bovine/ovine/porcine muscle, liver, kidney, fat (where authorized use applies); milk and eggs (where applicable); feed and medicated premixes; surface and wastewater near aquaculture sites.
Monitoring objectives include regulatory surveillance, import/export controls, producer QA/QC, and environmental exposure assessment.
Health Impact — Human Toxicity
Therapeutic use is generally well tolerated; common adverse effects include gastrointestinal symptoms, headache, dizziness, and fatigue, often related to parasite burden. At high doses, transient hepatic enzyme elevations can occur. Occupational exposure in laboratories should be minimized; handle as a pharmaceutical active with appropriate controls.
Risk to consumers is managed via adherence to MRLs and withdrawal periods; robust residue testing underpins consumer protection.
Environmental Impact — Fate and Effects on Wildlife
Praziquantel can enter aquatic environments via aquaculture treatments and effluents. It shows moderate hydrophobicity and is subject to biotransformation and photodegradation; environmental persistence is typically limited, but localized exposures may affect non-target invertebrates. Environmental monitoring focuses on water, sediment, and biota in and around treated facilities.
Use of sensitive analytical methods and matrix-matched calibration is recommended to characterize low-level environmental residues.
Safety Measures for Laboratory Handling
Handle in a fume hood or containment with appropriate PPE (lab coat, nitrile gloves, safety glasses). Avoid inhalation of dusts and skin contact. Use clean, dedicated tools to prevent cross-contamination. Dispose of waste according to local regulations for pharmaceutical actives.
Consult the substance safety data sheet (SDS) for detailed hazard and first-aid information.
Analytical Challenges
Co-extractives from high-fat tissues and complex aquaculture feeds can suppress ionization in LC-MS/MS. Enantiomeric composition and the presence of positional isomers/metabolites (e.g., hydroxylated species) require selective chromatographic separation. Low μg/kg regulatory limits necessitate high sensitivity and rigorous control of background contamination.
Sample Preparation
Common workflows include QuEChERS-style extraction with dispersive SPE cleanup, or LLE/SPE protocols optimized for moderately lipophilic pharmaceuticals. Enzymatic hydrolysis may be considered if residue definitions include conjugates. Use matrix-matched calibration and stable isotope-labelled internal standards to correct for recovery and matrix effects.
Analytical Methods
Primary: LC-MS/MS with ESI+ detection, employing reversed-phase chromatography. Confirmation via qualifier ion ratios and retention time tolerance. HPLC-UV can be used for higher concentration levels (e.g., formulation control) but lacks sensitivity for regulatory residues.
Method validation should cover selectivity, linearity, accuracy (recovery), precision, LOQ/LOD, matrix effects, and stability, following SANTE/ISO or equivalent guidelines.
Target Analytes and Metabolites
Typical residue targets include: Praziquantel (parent); hydroxy-praziquantel isomers (e.g., 4-hydroxylated metabolites). Laboratories should align analyte lists with the jurisdictional residue definition (parent compound and/or specific metabolites, free and/or conjugated forms).
Where stereoselective information is required, chiral LC may be employed to distinguish enantiomers.
Calibration and Traceability
Use certified reference materials for primary calibration and stable isotope-labelled internal standards for quantitation. Prepare calibration ranges covering expected MRLs and method LOQs. Ensure traceability of concentrations to SI units where applicable, supported by documentation and a certificate of analysis (CoA).
Storage and Stability
Store solid materials in tightly closed containers, protected from light and moisture, at recommended temperatures. Store solutions refrigerated or frozen as indicated on the CoA. Assess solution stability and perform periodic checks; use amber glass to mitigate photodegradation.
Product Portfolio
HPC Standards offers Praziquantel reference materials in neat form and ready-to-use solutions at various concentrations, along with key metabolites and stable isotope-labelled derivatives for internal standardization. Custom concentrations, solvent systems, and mixtures for multi-residue methods are available on request.
All products are produced under stringent quality controls, tested according to international quality requirements, and supplied with comprehensive documentation.
Typical Applications for Laboratories
Regulatory residue testing in aquaculture products; QA/QC in veterinary pharmaceutical manufacturing and feed medicated premixes; method development and validation (single- or multi-residue methods); proficiency testing and instrument performance verification; environmental monitoring of aquaculture sites.
Compliance and Documentation
Each reference material is delivered with a CoA detailing identity, purity, concentration (for solutions), uncertainty, storage conditions, and recommended shelf life. Batch-specific data support ISO-compliant validation and routine QA/QC workflows.
Ordering Information
Available pack sizes and concentrations suited to screening and confirmatory methods. Bulk and custom-pack options for high-throughput labs. Technical support provided by HPC Standards for method selection, stability guidance, and custom mixes.
Related Reference Materials
Complementary products include reference materials for other antiparasitic agents used in aquaculture and veterinary medicine (e.g., emamectin benzoate, levamisole, flubendazole, nitroxynil), plus their metabolites and stable isotope-labelled derivatives, enabling comprehensive residue surveillance.


