New user? / Forgot your password?
0 Item | 0,00 €
Fluralaner — Reference Materials for Residue and Veterinary Drug Analysis
Fluralaner Precision Reference Materials for Confident Residue Quantification
Ensure compliance with robust, ISO-aligned results HPC Standards supplies highpurity fluralaner reference materials, its key metabolites, and stable isotopelabelled analogues for LCMSMS. Our traceable neat and readytouse solutions deliver consistent calibration, low LOQs, and reliable recovery across challenging matrices such as eggs, poultry tissues, and highfat samples. Backed by comprehensive CoAs purity, identity, uncertainty and tailored blends for your residue definition and MRLs, we are your partner for food and environmental monitoring with proven quality and supply continuity.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
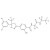 | 693110 | 1X10MG | Please log in. | |
ISO 17034 Certified Reference Material D4-Fluralaner solution | 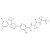 | 693111 | 1X1ML | Please log in. |
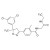 | 681292 | 1X10MG | Please log in. | |
Fluralaner solution | 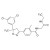 | 682118 | 1X10ML | Please log in. |
HPC Standards provides fit-for-purpose reference materials for fluralaner, its metabolites, and stable isotope‑labelled derivatives to support robust quantification in food and environmental matrices under international quality requirements.
Overview
Fluralaner is a systemic insecticide and acaricide (isoxazoline class) used in veterinary medicine for the control of fleas and ticks in companion animals and mites in poultry. It acts on ligand‑gated chloride channels and is administered orally or topically in pets and via drinking water in poultry.
Key identifiers: CAS 864731-61-3; molecular formula C22H17Cl2F6N3O3; molar mass 556.29 g/mol; IUPAC: 4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-1,2-oxazol-3-yl]-2-methyl-N{2-oxo-2-[(2,2,2-trifluoroethyl)amino]ethyl}benzamide. Synonyms/trade names include Bravecto (companion animals) and Exzolt (poultry).
Chemical Identity and Properties
Structural class: isoxazoline; chirality: racemic mixture. High halogenation (Cl, CF3 groups) confers lipophilicity and persistence. Typical bioavailability 20–27% (reduced fasted); elimination half‑life ~9–16 days in target species.
Analytical implications: non‑polar, thermally labile; routinely determined by LC–MS/MS. Expected strong response in ESI (positive or negative depending on method development); monitor halogenated fragment ions.
Mode of Action
Fluralaner inhibits GABA‑gated and L‑glutamate‑gated chloride channels in arthropods, producing neuroexcitation and death. Selectivity for insect and acarine channels underpins its veterinary use profile.
Uses and Applications
Companion animals: oral and topical products for fleas and ticks; extended duration formulations are common.
Poultry: drinking‑water solutions for mite (e.g., Dermanyssus gallinae) control. Use in production animals introduces the need for residue monitoring in eggs and meat.
Regulatory Status
Approved in the EU (veterinary use), US (Rx‑only for pets; additional indications approved over time), AU, CA and other regions. Poultry use approved in the EU for drinking‑water administration.
Maximum residue limits (MRLs)/tolerances for food‑producing species are established in many jurisdictions. Always consult current EU MRL database, US tolerances, and national compendia for the latest limits and residue definitions.
Residue Definitions and Target Matrices
Residue definitions may include parent fluralaner only or parent plus specified metabolites, depending on jurisdiction. Target matrices typically include poultry muscle, skin/fat, liver, and eggs; pet exposure studies may inform environmental assessments.
Ancillary matrices: feed, drinking water, litter/manure, and surface wipes (animal housing) for stewardship monitoring.
Monitoring and Surveillance
Compliance monitoring focuses on edible tissues and eggs post‑treatment, respecting withdrawal periods. Screening and confirmatory methods should meet identification criteria (e.g., SANTE/ISO) for ion ratios, retention time, and mass accuracy (where applicable).
Trend monitoring in integrated supply chains can be implemented via periodic batch testing and proficiency participation.
Sample Preparation and Extraction
Common workflows: protein precipitation (acetonitrile or methanol) followed by liquid–liquid partitioning and solid‑phase cleanup (e.g., C18, polymeric reversed phase). For complex matrices (eggs, fat), use fat removal strategies (hexane partitioning, EMR‑Lipid, or zirconia‑based sorbents).
QuEChERS variants (citrate/acetate buffered) with tailored d‑SPE cleanup are also used, particularly for multi‑residue pesticide methods in food of animal origin.
Analytical Methods (LC–MS/MS)
Typical platforms: UHPLC coupled to triple quadrupole MS/MS. Columns: C18 or phenyl‑hexyl, sub‑2 µm; gradient elution with aqueous ammonium formate/acetate and organic (MeOH/ACN). Optimize collision energies for halogen‑rich fragments; confirm with two transitions where applicable.
Method performance targets: LOQs at or below one‑half MRL; recovery 70–120% with RSD ≤20% across matrices; matrix‑matched or isotopically labelled calibration to correct ion suppression.
Quality Control and Reference Materials
HPC Standards supplies traceable reference materials for fluralaner, relevant metabolites, and stable isotope‑labelled analogues for internal standardisation. Certificates of Analysis include purity, identity (NMR/HRMS), and uncertainty per ISO‑aligned procedures.
Available formats: neat materials and ready‑to‑use solutions at validated concentrations for routine calibration, system suitability, and proficiency testing support.
Storage and Stability
Store neat materials tightly sealed at 2–8 °C or per CoA guidance, protected from light and moisture. Solution standards are typically stable refrigerated; assess long‑term stability and assign expiry based on verified studies. Avoid repeated freeze–thaw cycles; use aliquots.
Calibration and Measurement Uncertainty
Use multi‑level matrix‑matched calibration or isotope dilution with 13C/2H‑labelled internal standards to reduce bias. Establish intermediate precision across days and analysts; include bracketing checks and independent QC levels.
Combine contributions from purity, weighing, volumetry, and calibration model to document measurement uncertainty in line with ISO/IEC 17025 expectations.
Safety and Handling
Handle fluralaner reference materials in a chemical fume hood with appropriate PPE (lab coat, nitrile gloves, eye protection). Avoid dust and aerosols. Dispose of waste according to local regulations for halogenated organics.
Emergency measures: in case of skin/eye contact, rinse with water; seek medical advice as required. Refer to the product SDS for detailed hazard information.
Occupational Exposure and Human Toxicity
Fluralaner targets arthropod chloride channels; human acute toxicity is low to moderate based on available veterinary safety data, but ingestion or significant exposure should be avoided. Symptoms of overexposure may include gastrointestinal or neurological effects; treat symptomatically.
Implement good laboratory practice to minimise exposure; no occupational exposure limit values are universally established—follow institutional risk assessments.
Environmental Fate
Due to halogenation and lipophilicity, fluralaner can exhibit persistence and sorption to organic matter. Excreted residues from treated animals may enter manure and the environment; degradation depends on conditions (aerobic/anaerobic, photolysis).
Consider manure management and wastewater controls to mitigate environmental release, especially in intensive poultry operations.
Ecotoxicology and Effects on Wildlife
Fluralaner is highly active against arthropods; potential risks to non‑target invertebrates should be considered where environmental exposure may occur. Risk assessments should address aquatic invertebrates and beneficial arthropods near application sites.
Follow label stewardship to protect non‑target species and comply with local environmental regulations.
Cross‑Reactivity and Analytical Interferences
Isoxazoline co‑formulants or structurally related compounds (e.g., fipronil in multi‑residue screens) may co‑elute or share fragments. Resolve chromatographically and confirm with qualifier ions and retention time matching.
Mitigate matrix effects by using isotope‑labelled internal standards and appropriate cleanup.
Metabolites and Isotopically Labelled Standards
Where residue definitions include metabolites, ensure coverage with dedicated reference materials. Isotopically labelled fluralaner internal standards improve accuracy, particularly in high‑fat matrices.
HPC Standards offers bespoke mixtures tailored to your residue definition and MRLs.
Compliance and Documentation
Methods should align with recognized guidelines (e.g., EU SANTE/2023 series, Codex, FDA/USDA) for validation and identification criteria. Maintain full traceability of reference materials, calibration records, and QC charts for audits.
HPC Standards supports documentation needs with comprehensive CoAs, stability statements, and batch traceability.
Analytical Standards
Our portfolio includes: fluralaner reference materials (neat and solution), metabolite standards (where applicable), and stable isotope‑labelled internal standards for LC–MS/MS quantification. Custom concentrations and multi‑analyte blends are available on request.
All products are produced and tested according to international quality requirements to meet the highest industrial standards for food and environmental analysis.


