New user? / Forgot your password?
0 Item | 0,00 €
Hydroxybenzoic acid – Reference Materials for Reliable Quantification
Hydroxybenzoic acid Precision Reference Materials for Confident Compliance
Accelerate your LCGC workflows with ISO-aligned, traceable Hydroxybenzoic acid standards from HPC Standards. Calibrate and control isomer-specific quantification 2-, 3-, 4-hydroxybenzoic acid in food, environmental, and consumer-product matrices with ready-to-use certified solutions or high-purity neat materials. Deuterated internal standards, rigorous CoAs, and validated uncertainty ensure robust separation, linearity, and reproducible recoveriesso your lab meets regulatory limits with confidence, audit readiness, and uninterrupted supply. Custom concentrations, solvent systems, and multi-analyte mixes available on request.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 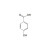 | 683351 | 1X1000MG | Please log in. |
ISO 17034 Certified Reference Material | 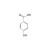 | 694265 | 1X500MG | Please log in. |
ISO 17034 Reference Material |  | 682526 | 1X1000MG | Please log in. |
ISO 17034 Certified Reference Material | 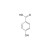 | 694316 | 1X500MG | Please log in. |
Ensure confident identification and quantification of hydroxybenzoic acid isomers with ISO-compliant reference materials for food, environmental, and consumer-product testing.
Overview
Hydroxybenzoic acid refers to monohydroxylated benzoic acids, most commonly the positional isomers 2-hydroxybenzoic acid (salicylic acid), 3-hydroxybenzoic acid, and 4-hydroxybenzoic acid (p-hydroxybenzoic acid). These phenolic acids occur naturally in plants, arise as transformation products of parabens and other aromatics, and are used as building blocks in pharmaceuticals and polymers. They are routinely monitored in food, environmental, and consumer-product matrices.
Accurate residue analysis requires isomer-specific separation and calibration using traceable reference materials to control matrix effects and ensure method robustness.
Chemical Identity and Isomers
Hydroxybenzoic acid (molecular formula C7H6O3; MW ≈ 138.12 g/mol) comprises three principal positional isomers: 2-hydroxybenzoic acid (salicylic acid; CAS 69-72-7), 3-hydroxybenzoic acid (CAS 99-06-9), and 4-hydroxybenzoic acid (CAS 99-96-7). The isomeric position influences acidity, hydrogen bonding, solubility, chromatographic behavior, and toxicological profiles, necessitating isomer-resolved analysis.
Physicochemical Properties
Weak aromatic carboxylic acids with a carboxyl pKa around 2.9–3.0 and a phenolic pKa near 9–10 (isomer-dependent). Moderate water solubility that increases with pH; low to moderate hydrophobicity (low logP) and strong UV absorbance due to the aromatic ring. In LC-MS, they readily form deprotonated ions [M−H]− (m/z 137) under negative ESI.
Occurrence and Sources
Natural constituents of many fruits, grains, and botanicals; microbial and plant metabolism products; degradation products of parabens (p-hydroxybenzoic acid esters) and certain aromatic pollutants; processing and storage can modify levels in foods and beverages (e.g., fermentation).
Uses and Industrial Relevance
Salicylic acid is a key pharmaceutical intermediate and active for keratolytic applications; p-hydroxybenzoic acid is a precursor to parabens and specialty polymers; hydroxybenzoic acids serve as reagents, intermediates, and functional additives in various sectors. Their presence in products and the environment drives routine compliance monitoring.
Regulatory Landscape
Regulatory oversight depends on context and region. Examples include: cosmetic use restrictions for salicylic acid and paraben derivatives (EU Cosmetic Regulation (EC) No 1223/2009 and analogous frameworks); substance registration and safety data requirements under REACH and comparable schemes; product-specific specifications or migration limits in food-contact materials; environmental permitting for manufacturing effluents. Laboratories should verify the latest regional requirements and client specifications.
Monitoring and Target Matrices
Typical matrices include drinking and surface waters, wastewater, soils and sediments, plant materials and foods (grains, fruits, beverages), cosmetics and personal care products, pharmaceutical formulations, and industrial process streams. Method development must address matrix complexity, potential conjugates (e.g., glucuronides), and isomer co-elution.
Methods of Analysis
LC-UV/DAD and LC-MS/MS are preferred for specificity and sensitivity; HILIC or reversed-phase LC with isocratic or gradient elution separates isomers effectively. GC-MS is feasible following derivatization (e.g., silylation) to increase volatility and thermal stability. FTIR or NMR may support identity confirmation for pure materials. Quantification relies on external or internal calibration with matrix-matched standards and isotopically labelled internal standards where available.
Sample Preparation and Cleanup
Common approaches include dilute-and-shoot for simple aqueous matrices, solid-phase extraction (reversed-phase or mixed-mode anion exchange) for enrichment and cleanup, protein precipitation for biological samples, and QuEChERS-type protocols for food matrices adapted to acidic phenolics. Maintain pH control to keep analytes in the desired ionization state, minimize phenolic oxidation, and avoid ester hydrolysis for paraben-related studies.
Human Toxicity
Hydroxybenzoic acids exhibit low to moderate acute toxicity, with isomer-dependent profiles. Salicylic acid can cause skin and eye irritation and, at elevated systemic exposures, salicylate-related effects (e.g., metabolic and gastrointestinal disturbances). p-Hydroxybenzoic acid is generally of low acute toxicity but may irritate eyes/skin. Standard laboratory hygiene and exposure controls mitigate risk.
Environmental Impact and Effects on Wildlife
Hydroxybenzoic acids are typically of low bioaccumulation potential and are readily biodegradable under aerobic conditions; degradation kinetics depend on matrix and microbial activity. Acute aquatic toxicity is generally low to moderate. Transformation from consumer-product residues (e.g., parabens) can lead to environmental occurrence; monitoring helps evaluate source control and treatment efficacy.
Safety Measures and Handling
Implement standard chemical hygiene: wear lab coat, gloves, and eye protection; handle powders to minimize dust and inhalation; avoid contact with skin and eyes. Ensure adequate ventilation and use fume hoods for weighing and solution preparation. In case of contact, rinse affected area with water; consult the safety data sheet for first aid and spill response.
Storage and Stability
Store solid materials tightly sealed, dry, and protected from light at controlled room temperature or as specified on the certificate. Store solutions refrigerated (2–8 °C) unless otherwise stated. Use amber containers to prevent photodegradation of phenolic compounds. Allow solutions to equilibrate to room temperature before opening to avoid condensation; record open-date and adhere to assigned shelf-life and stability statements.
Analytical Reference Materials
HPC Standards provides traceable reference materials for 2-, 3-, and 4-hydroxybenzoic acid to support method development, system suitability, calibration, and quality control. Formats include neat materials and ready-to-use certified solutions at defined concentrations in high-purity solvents. Isotopically labelled analogues (e.g., deuterated p-hydroxybenzoic acid) are available for internal standardization to correct for matrix effects and recovery.
Each reference material is accompanied by a Certificate of Analysis detailing identity confirmation, purity, uncertainty, solvent composition (for solutions), and traceability to international standards.
Quality and Accreditation
Our reference materials are produced under stringent quality systems aligned with international requirements to ensure consistency, traceability, and long-term reliability. Batches are tested with validated methods, and measurement uncertainty is rigorously estimated. Documentation supports audit readiness for regulated laboratories.
Applications in Food and Environmental Analysis
Food: profiling of phenolic acids, verification of natural occurrence versus additive-related sources, monitoring of paraben metabolism products, and authenticity studies. Environmental: surveillance of wastewater and surface waters for consumer-product residues and transformation products; assessment of treatment efficacy; source apportionment. Consumer products: specification testing in cosmetics and personal care formulations.
Method Performance Targets
Typical objectives include LOQs in the low µg/L (water) to low mg/kg (solids) range, isomer resolution with baseline separation under routine gradients, linear calibration over 2–3 orders of magnitude, recoveries of 70–120% depending on matrix, and RSDs meeting internal QC criteria. Reference materials enable ongoing verification via system suitability and QC charts.
Procurement and Customization
Standard pack sizes and concentrations are available for immediate dispatch. Custom concentrations, solvent systems, ampoule or vial formats, and multi-analyte mixes (including related phenolics or paraben suites) can be prepared on request to match your method and instrumentation. Long-term supply and lot reservation ensure continuity for validated methods.


