New user? / Forgot your password?
0 Item | 0,00 €
Delafloxacin
Ensure Accurate Residue Analysis with High-Purity Delafloxacin Reference Materials
HPC Standards GmbH offers high-purity Delafloxacin reference materials, essential for precise residue analysis in food and environmental samples. Our products meet the highest industrial standards, supporting laboratories in achieving compliance with regulatory limits. Trust HPC Standards for reliable and accurate analytical solutions.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 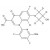 | 692714 | 1X5MG | |
ISO 17034 Certified Reference Material D5-Delafloxacin solution | 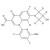 | 692715 | 1X1ML | |
ISO 17034 Certified Reference Material Delafloxacin solution | 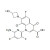 | 690481 | 1X1ML | |
ISO 17034 Reference Material | 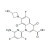 | 692599 | 1X25MG |
High-purity reference materials for accurate residue analysis in food and environmental samples
Overview
Delafloxacin, sold under the brand name Baxdela among others, is a fluoroquinolone antibiotic used to treat acute bacterial skin and skin structure infections (ABSSSI) and community-acquired bacterial pneumonia (CABP) in adults. It was developed by Melinta Therapeutics and approved by the FDA in June 2017.
Uses
Delafloxacin is indicated for the treatment of adults with ABSSSI caused by designated susceptible bacteria including Gram-positive organisms such as Staphylococcus aureus (MRSA and MSSA), Staphylococcus haemolyticus, and Gram-negative organisms such as Escherichia coli and Pseudomonas aeruginosa. It is also used to treat CABP caused by susceptible bacteria like Streptococcus pneumoniae and Haemophilus influenzae.
Regulatory
Delafloxacin is available by prescription only (℞) in the United States, Canada, and the European Union. The FDA approved Delafloxacin for ABSSSI and CABP in June 2017 after it was shown to be noninferior to vancomycin plus aztreonam in clinical trials.
Monitoring
The FDA has mandated a 5-year surveillance study to monitor the emergence of resistance to Delafloxacin. Additionally, studies on the IV form in pregnant rats were required to determine distribution to the reproductive tract.
Health Impact
Delafloxacin, like other fluoroquinolones, carries a black box warning about the risk of tendinitis, tendon rupture, peripheral neuropathy, central nervous system effects, and exacerbation of myasthenia gravis. Adverse effects reported in clinical trials include nausea, diarrhea, headache, elevated transaminases, and vomiting.
Human Toxicity
Delafloxacin has not been tested in pregnant women. It is excreted 65% through urine and 28% via feces, mostly in unmetabolized form. Clearance is reduced in individuals with severe kidney disease.
Environmental Impact
No specific data on the environmental impact of Delafloxacin is available; however, as with all pharmaceuticals, proper disposal and environmental monitoring are recommended to prevent contamination.
Effects on Wildlife
There is limited information on the effects of Delafloxacin on wildlife. Environmental monitoring and controlled disposal are advised to minimize potential impacts.
Safety Measure
Delafloxacin should be used with caution due to its potential for serious side effects. Patients should be monitored for signs of tendonitis, tendon rupture, and other adverse effects. It should not be used with certain antacids, dietary supplements, or drugs buffered with divalent or trivalent cations.
Regulation
Delafloxacin is regulated as a prescription-only medication in various regions including the US, EU, and Canada. It is subject to ongoing surveillance studies to monitor resistance and safety.
Analytical Standards
HPC Standards GmbH provides high-purity reference materials for Delafloxacin, ensuring accurate and reliable residue analysis. Our products meet the highest industrial standards and are essential for laboratories conducting food and environmental analysis to ensure compliance with regulatory limits.


