New user? / Forgot your password?
0 Item | 0,00 €
Josamycin Reference Materials
Josamycin Precision Reference Materials for Confident LCMSMS Quantification
Ensure reliable residue analysis with HPC Standards traceable Josamycin reference materials. Our high-purity neat substances and ready-to-use certified solutions support robust method development, matrix-matched calibration, and routine QC across food and environmental samples. Delivered with a comprehensive CoA purity, uncertainty, stability and manufactured to international quality requirements, these materials enable ISOIEC 17025-compliant validation and SANTE-aligned confirmation. Benefit from consistent batch-to-batch performance, tailored concentrations, and temperature-controlled shippingyour dependable partner for MRL compliance and antimicrobial surveillance.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
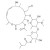 | 676995 | 1X50MG | Please log in. | |
Josamycin solution 10 µg/ml Acetone | 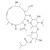 | 689078 | 1X10ML | Please log in. |
High-quality josamycin reference materials for robust method development, calibration, and regulatory residue monitoring in food and environmental matrices.
Overview
Josamycin is a macrolide antibiotic produced by Streptomyces species. It exhibits activity primarily against Gram-positive bacteria, atypical pathogens (e.g., Mycoplasma, Chlamydia), and select Gram-negative organisms. In residue and environmental analysis, josamycin is a key target as a veterinary medicinal product and potential environmental contaminant.
HPC Standards provides traceable josamycin reference materials designed for quantitative analysis by LC–MS/MS and related chromatographic techniques across complex matrices.
Chemical and Physical Properties
Class: 16-membered macrolide antibiotic; amphiphilic lactone ring with multiple glycosidic substituents.
Typical behavior: Limited thermal stability at elevated temperatures; susceptible to degradation under strong acidic or basic conditions; moderate lipophilicity; low volatility.
Analytical implications: Requires controlled pH, protection from light and moisture, and use of appropriate organic solvents to maintain integrity during analysis and storage.
Uses and Applications
Clinical and veterinary antibiotic for respiratory tract, skin/soft-tissue, and atypical infections depending on jurisdictional approvals.
Analytical relevance: Monitoring residues in food of animal origin, surveillance of antimicrobial occurrence in wastewater and surface waters, and studies of antimicrobial resistance drivers in environmental compartments.
Regulatory and MRLs
Residues of josamycin in food-producing animals are regulated by jurisdiction-specific frameworks (e.g., EU: Regulation (EC) No 470/2009 and Commission Regulation (EU) No 37/2010; US: FDA tolerances; Codex where applicable). Maximum residue limits (MRLs) vary by species, tissue, and region.
Recommendation: Consult the latest official MRL databases and national legislation to confirm current limits and authorized uses before method validation and reporting.
Monitoring and Target Matrices
Food: Muscle, liver, kidney, fat, milk, eggs, and honey for veterinary drug surveillance.
Environmental: Wastewater influent/effluent, surface water, sediments, and agricultural soils near livestock operations.
Scope: Targeted LC–MS/MS monitoring within national residue control plans, screening studies for antimicrobial pollution, and confirmatory analyses in enforcement laboratories.
Analytical Methods
Preferred technique: LC–MS/MS (MRM) for high selectivity and sensitivity in complex matrices. Use reversed-phase columns (C18 or polar-embedded) with gradient elution and volatile buffers (e.g., formic or acetic acid with ammonium formate).
Ionization: Positive ESI; monitor multiple qualifier/quantifier transitions and ion ratios for confirmation per SANTE/ISO guidance.
Alternatives: HPLC–UV/FLD as screening in clean matrices, though generally less sensitive and selective than MS-based detection.
Sample Preparation
Typical workflows: Protein precipitation or liquid–liquid extraction for milk; SPE (e.g., HLB, mixed-mode) for cleanup in meat, liver, kidney, and eggs; QuEChERS-based approaches adapted for veterinary drugs with dispersive cleanup; SPE or online-SPE for aqueous environmental samples.
Key controls: Stabilize pH (mildly acidic), minimize exposure to light/heat, and include matrix-matched calibration and internal standards to mitigate matrix effects.
Reference Materials from HPC Standards
Offerings: Neat josamycin reference materials and ready-to-use certified solutions at defined concentrations for immediate calibration and QC.
Quality: Manufactured under stringent quality requirements with full documentation (Certificate of Analysis, purity, uncertainty, and stability data). Suitable for method development, system suitability, calibration curves, recovery studies, and proficiency testing support.
Calibration and Quantification
Use multi-level calibration spanning expected MRLs/action limits and environmental concentrations. Apply matrix-matched or standard addition approaches for complex matrices.
Employ internal standards for correction of extraction efficiency and ion suppression. Where available, stable isotope-labelled derivatives are preferred for highest accuracy.
Quality Assurance and Documentation
Each reference material is delivered with a detailed Certificate of Analysis including identity confirmation, purity assay, concentration (for solutions), uncertainty, and recommended storage conditions.
Traceability and batch consistency support ISO/IEC 17025 method validation, routine QC (blanks, spikes, duplicates), and compliance with SANTE/12682 and related guidance for confirmatory methods.
Health Impact – Human Toxicity
Pharmacology: Protein synthesis inhibitor (50S ribosomal subunit). As residues, potential for allergic reactions or gastrointestinal disturbance in sensitive individuals; risk is controlled through adherence to MRLs and withdrawal periods.
Occupational exposure: Avoid inhalation and dermal contact with powders/solutions; implement lab hygiene and PPE as per SDS.
Environmental Impact – Effects on Wildlife and Microbiota
Ecotoxicology: Macrolides can affect aquatic microbial communities and may contribute to selection pressure for antimicrobial resistance. Acute toxicity to higher trophic levels is generally lower than to microorganisms, but chronic effects are matrix- and concentration-dependent.
Risk management: Waste minimization and appropriate disposal reduce environmental loading and resistance development.
Safety Measures and Handling
Handle in a fume hood with lab coat, nitrile gloves, and eye protection. Avoid aerosol formation. Use amber glassware to protect from light.
Consult the Safety Data Sheet for hazard classifications, first aid, and spill response. Dispose of waste according to institutional and regulatory requirements for pharmaceutical substances.
Storage and Stability
Neat material: Store tightly closed at 2–8 °C or frozen (≤ −20 °C) in a dry, dark environment. Protect from moisture and strong acids/bases.
Solutions: Store at low temperature in suitable solvents (e.g., acetonitrile/water or methanol) in amber vials; verify stability and establish replacement intervals based on CoA guidance.
Related Compounds and Isotope-Labelled Materials
Associated analytes: Other macrolides (e.g., erythromycin, spiramycin, tilmicosin, tylosin) may be co-monitored in multi-residue methods.
Stable isotope-labelled internal standards for josamycin or structurally related macrolides can improve quantification accuracy; availability may vary—contact us for current portfolio.
Industries and Use Cases
Food safety laboratories, contract research organizations, regulatory and official control labs, environmental monitoring facilities, and academic research groups focused on antimicrobial residues and resistance.
Applications include MRL compliance testing, surveillance studies, method validation, and proficiency testing support.
Ordering, Packaging, and Shipping
Products are supplied in tamper-evident amber glass vials with clear labeling and batch identification. Temperature-controlled shipping is available where required to maintain stability.
Custom concentrations, mixed calibrants, and bulk quantities are available on request to streamline your workflows and reduce preparation errors.


