New user? / Forgot your password?
0 Item | 0,00 €
Benzylidene camphor – Reference materials for residue analysis
Benzylidene camphor HighPurity Reference Materials for Confident Residue Analysis
Ensure accuracy, traceability, and regulatory compliance with HPC Standards benzylidene camphor reference materials. Designed for environmental and product testing, our rigorously characterized neat materials and gravimetric solutions support LCMSMS, HPLCUV, and GCMS workflows across complex matriceswastewater, surface waters, sediments, sludge, biota, and cosmetics. Benefit from batch-specific CoAs purity, identity, uncertainty, custom UVfilter mixes incl. 3BC, 4MBC, and technical support for method development, matrix challenges, and QAQC. Tested to international quality requirements, our standards help your laboratory meet accreditation needs and deliver reproducible results.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
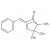 | 693834 | 1X10MG | ||
Benzylidene camphor solution | 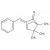 | 693835 | 1X5ML |
High-purity benzylidene camphor reference materials for reliable identification, quantification, and regulatory compliance in environmental and product testing.
Overview
Benzylidene camphor denotes a group of camphor-derived aromatic compounds widely used as organic UV filters in cosmetics and personal care products. In residue analysis, benzylidene camphor and selected derivatives are monitored in surface waters, wastewater, sediments, sludge, and biota due to their persistence and potential endocrine activity. Laboratories require rigorously characterized reference materials to ensure data integrity across complex matrices.
Chemical identity and synonyms
Benzylidene camphor refers to camphor bearing a benzylidene moiety; common analytical targets include specific derivatives. Frequently encountered names include 3-benzylidene camphor (3-BC) and 4-methylbenzylidene camphor (4-MBC, enzacamene), among others. Exact isomer specification is critical for method development and quantification.
Note: Use unambiguous identifiers (CAS No., IUPAC name, isomer position) in LIMS and reporting to avoid misclassification of closely related compounds.
Physicochemical properties
- Aromatic, hydrophobic UV-absorbing compounds with relatively low water solubility and high affinity for organic matter.
- Moderate to high log Kow values lead to enrichment in sediments, sludge, and biota.
- Light-absorbing chromophores may undergo phototransformation; however, some derivatives exhibit notable photostability under use conditions.
Uses
- UV filters in cosmetic formulations (e.g., sunscreens, day creams, lip care) for UVB/UVA protection.
- Potential use as UV stabilizers in certain materials. Analytical monitoring focuses on consumer-product residues and environmental release via wastewater pathways.
Occurrence and target matrices
- Environmental: influent/effluent wastewater, surface waters, sediments, sewage sludge, stormwater, and marine/coastal waters.
- Biota: fish tissue, invertebrates, and biofilms due to bioaccumulation potential.
- Product control: cosmetic finished products and raw materials for specification testing and market surveillance.
Regulatory status
- Regulatory frameworks differ by jurisdiction. In the EU and other regions, certain benzylidene camphor derivatives (e.g., 3-BC, 4-MBC) have been subject to restrictions or non-approval in cosmetic products following safety assessments.
- Environmental monitoring requirements may be set at national or regional levels for organic UV filters discharged via wastewater treatment plants.
- Laboratories should verify current listings (e.g., cosmetics positive/negative lists, priority substances, local discharge limits) for the specific derivative(s) under investigation.
Monitoring and surveillance
- Routine screening in wastewater and receiving waters near densely populated or touristic areas.
- Trend monitoring in sediments and sludge to evaluate long-term accumulation.
- Product compliance testing to confirm ingredient specifications and labeling claims.
Analytical methods
- LC–MS/MS: Preferred for sensitive, selective quantification across water and biota matrices; supports multi-residue panels of UV filters.
- HPLC–DAD/UV: Suitable for screening and product control where higher concentrations are expected.
- GC–MS(/MS): Applicable following derivatization for more volatile targets or when harmonized with existing workflows.
- HRMS: Useful for suspect/non-target screening of transformation products and co-occurring UV filters.
Sample preparation
- Aqueous matrices: Solid-phase extraction (SPE; e.g., C18, polymeric, mixed-mode) with isotope-dilution or matrix-matched calibration.
- Solids/sediments/sludge: Accelerated solvent extraction (ASE), Soxhlet, or ultrasonic extraction with lipid cleanup (e.g., GPC, silica/alumina).
- Biota: Homogenization, solvent extraction, lipid removal, and cleanup to minimize matrix effects.
- Cosmetics: Solvent dilution or extraction in suitable organic media, filtration, and HPLC/LC–MS analysis.
Calibration and quantification
- Use matrix-matched calibration or isotope dilution where available to correct for recovery and ion suppression.
- Establish method detection/quantification limits appropriate for environmental and product-compliance objectives.
- Include procedural blanks, duplicates, and fortified samples for ongoing performance verification.
Reference materials
- HPC Standards provides benzylidene camphor reference materials to support method development, calibration, and QA/QC in environmental and product testing.
- Formats: neat materials and gravimetrically prepared solutions upon request, accompanied by a comprehensive certificate of analysis (purity, identity, uncertainty, storage, and handling guidance).
- Custom mixes: Multi-analyte blends for UV filter panels tailored to your target list and concentration ranges.
Quality assurance, traceability, uncertainty
- Each lot is tested according to international quality requirements to ensure identity, purity, and homogeneity.
- Traceability is established via purity assignment and gravimetric preparation, with stated measurement uncertainty to support accreditation needs.
- Batch-specific documentation enables full audit trails and reproducibility across studies and sites.
Health impact – human toxicity
- Exposure in the general population is primarily via dermal application of cosmetic products containing specific derivatives; systemic exposure depends on formulation and use patterns.
- Safety assessments for certain benzylidene camphor derivatives have raised concerns related to endocrine activity; regulatory outcomes vary by region and compound.
- Laboratory handling: avoid inhalation and skin/eye contact; follow institutional chemical safety procedures.
Environmental impact – fate and effects on wildlife
- Entry pathways include wash-off from consumers to wastewater and incomplete removal in treatment plants.
- Potential for bioaccumulation and sub-lethal effects has been reported for some derivatives in fish and invertebrates, including endocrine-related endpoints.
- Photolysis and biodegradation may occur but can be limited; sorption to sediments and sludge is common due to hydrophobicity.
Safety measures and handling
- Handle reference materials in a fume hood with appropriate PPE (lab coat, nitrile gloves, safety glasses).
- Avoid light exposure during preparation and storage; use amber glassware and minimize sample residence time in autosamplers.
- Dispose of waste according to institutional and regulatory requirements for organic solvents and hydrophobic organics.
Storage and stability
- Store sealed containers at 2–8 °C or as indicated on the certificate; protect from light and moisture.
- Allow solutions to equilibrate to room temperature before opening to prevent condensation; re-cap promptly to limit volatilization and oxidation.
- Monitor expiration dates and requalification intervals; verify stability in working solutions under laboratory conditions.
Related compounds and metabolites
- Common co-occurring analytes: 4-methylbenzylidene camphor (4-MBC), 3-benzylidene camphor (3-BC), octocrylene, benzophenones, and other cosmetic UV filters.
- Transformation products may form via photolysis or biotransformation; high-resolution MS workflows support suspect screening when required.
Applications for laboratories
- Environmental monitoring programs assessing UV filter loads and temporal trends.
- Wastewater treatment plant studies (influent/effluent removal, sludge partitioning).
- Product control and reformulation support for cosmetic manufacturers and raw material suppliers.
- Method validation, interlaboratory comparisons, and proficiency testing using traceable reference materials.
Documentation and support
- Each reference material is delivered with a certificate of analysis and detailed handling instructions.
- Technical support available for method setup (LC–MS/MS, HPLC–UV, GC–MS), matrix challenges, and custom blends aligned to your regulatory scope.


