New user? / Forgot your password?
0 Item | 0,00 €
Aspartic Acid – Reference Materials for Accurate Quantification
Aspartic acid High-Purity Reference Materials for CONFIDENT Quantification
Accelerate your amino acid analytics with HPC Standards Aspartic acid reference materials and 13C15Nlabelled derivatives. Achieve traceable calibration, robust method validation, and consistent QC across LCMSMS, HPLCUPLC, GCMS derivatized, and CE workflows. Supplied with comprehensive CoA, SI traceability, and SDS, our neat and solution RMs deliver batch-to-batch reliability for food, pharma, biomedical, and environmental labs. Custom mixes, matrix-matched standards, and technical support available to fit your method and regulatory needs.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
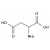 | 691096 | 1X100MG | ||
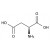 | 690766 | 1X100MG | Please log in. | |
ISO 17034 Certified Reference Material L-Aspartic acid solution | 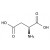 | 694011 | 1X5ML | Please log in. |
HPC Standards provides high-purity aspartic acid reference materials and stable isotope–labelled derivatives for reliable calibration, method validation, and quality control in food, pharmaceutical, biomedical, and environmental laboratories.
Overview
Aspartic acid (L-aspartic acid; CAS 56-84-8) is a proteinogenic, acidic amino acid with side-chain carboxyl functionality. Formula: C4H7NO4; molecular weight: 133.10 g/mol. Key acid-base properties: pKa1 ≈ 2.1 (α-COOH), pKa2 ≈ 3.9 (side-chain COOH), pKa3 ≈ 9.8 (α-NH3+); isoelectric point ≈ 2.8. It appears as a white crystalline solid, is sparingly soluble in water, and forms more soluble salts (e.g., sodium/potassium aspartate). Reference materials enable traceable quantification of free aspartic acid and total aspartate after hydrolysis.
Uses
- Calibration and system suitability for amino acid profiling by LC-MS/MS, HPLC (pre/post-column derivatization), ion-exchange chromatography, and capillary electrophoresis.
- Internal standardization (using 13C/15N-labelled aspartic acid) in targeted metabolomics, proteomics, and clinical chemistry.
- Quality control of protein hydrolysates, fermentation broths, dietary supplements, parenteral nutrition mixtures, and cell culture media.
- Food authenticity testing, protein digestibility studies, stability studies of biologics, and bioprocess monitoring.
Regulatory & Compliance
- Chemicals management: subject to EU REACH and CLP; aspartic acid is generally not classified as hazardous under GHS when pure and uncontaminated (verify current SDS).
- Applicable quality frameworks for analytical use include ISO/IEC 17034 (reference material production), ISO/IEC 17025 (testing), and ISO 9001 (quality management).
- Method validation should follow ICH Q2(R2)/Q14, ISO 17025 guidance, and sector-specific SOPs (food, pharma, clinical).
Monitoring
- Matrices: plasma/serum, urine, cerebrospinal fluid, tissues, cell lysates, bioreactor media, protein hydrolysates, food ingredients, and wastewater/effluents.
- Typical workflows: protein precipitation or SPE, derivatization (e.g., OPA/FMOC, AQC, PITC) for HPLC/CE; direct or mild derivatization for LC-MS/MS; GC-MS requires suitable silylation/esterification.
- Use matrix-matched calibration and isotopically labelled internal standards to correct for recovery and ion suppression.
Health Impact — Human Toxicity
- Aspartic acid is a natural dietary amino acid with low acute toxicity at typical exposure levels. It is widely present in proteins and metabolic pathways.
- Occupational exposure: dust may cause mechanical irritation to eyes, skin, and respiratory tract. No known chronic systemic toxicity at occupational levels when handled properly.
- Follow SDS for handling; avoid inhalation of powders and contact with eyes. Not intended for therapeutic use.
Environmental Impact — Effects on Wildlife
- Aspartic acid is readily biodegradable and exhibits low bioaccumulation potential. Aquatic toxicity is expected to be low relative to many industrial chemicals.
- As a naturally occurring amino acid, environmental persistence is minimal under normal conditions; standard wastewater treatment effectively reduces concentrations.
Safety Measures
- Personal protection: lab coat, safety glasses, and suitable gloves; use dust control and local exhaust ventilation when weighing powders.
- Handling: minimize dust generation, use antistatic measures, and work in clean, dry conditions.
- First aid: rinse eyes/skin with water upon contact; move to fresh air if inhaled; consult SDS for detailed guidance.
Analytical Methods
- LC-MS/MS: preferred for sensitivity and selectivity; use 13C/15N-aspartic acid as internal standard; aqueous or mild derivatization workflows.
- HPLC/UPLC: ion-exchange with ninhydrin detection; reverse-phase with pre-column derivatization (AQC, OPA/FMOC, PITC); UV/FLD detection.
- GC-MS: requires derivatization (e.g., TBDMS, MTBSTFA, or ethyl chloroformate protocols) for volatility.
- CE: background electrolyte optimization with derivatization enhances UV/FLD detectability.
Method Performance & Validation
- Establish linearity across expected ranges (e.g., low ng/mL to high μg/mL depending on matrix).
- Assess matrix effects, recovery, precision, trueness, LOD/LOQ, and robustness per ICH Q2(R2)/ISO 17025.
- Use certified reference materials and isotopically labelled internal standards to demonstrate traceability and measurement uncertainty.
Analytical Reference Materials from HPC Standards
- Neat materials: L-aspartic acid crystalline reference materials, assay and purity characterized with full documentation.
- Solution reference materials: gravimetrically prepared concentrations in water or water/acetonitrile, with stated uncertainty and stability data.
- Isotope-labelled options: 13C- and/or 15N-labelled L-aspartic acid for internal standardization in LC-MS/MS.
- Custom matrices and mixes: tailored amino acid panels, matrix-matched RMs (e.g., serum, culture media), and custom concentrations on request.
Certification & Documentation
- Each lot supplied with Certificate of Analysis (CoA) detailing purity, identity (NMR/IR/MS), water/volatiles, residual solvents (if applicable), and uncertainty.
- Traceability to SI where applicable; gravimetric preparation with calibrated balances and verified solvent purity.
- Accompanied by Safety Data Sheet (SDS) and recommended storage/handling instructions.
Storage & Stability
- Neat material: store tightly closed in a dry, cool place (2–8 °C), protected from moisture and light.
- Solutions: store refrigerated (2–8 °C) or frozen (≤ −20 °C) depending on matrix; avoid repeated freeze–thaw cycles; note assigned shelf-life on label/CoA.
- Allow to equilibrate to room temperature before opening to prevent moisture ingress; record opening date for inventory control.
Quality & Accreditation
- Manufactured under stringent quality management aligned with international standards for reference materials.
- Batch-to-batch consistency, homogeneity testing, and stability monitoring ensure reliable performance across shelf-life.
- Suitable for audits and regulatory submissions requiring documented measurement traceability.
Applications in Food & Environmental Analysis
- Nutritional profiling of foods and feeds (free and total amino acids); verification of fortification and formulation.
- Bioprocess control (e.g., amino acid consumption/production in fermentation); wastewater monitoring for nitrogenous metabolites.
- Support for proteomic workflows and peptide mapping via hydrolysis controls and calibration.
Packaging & Ordering
- Common pack sizes: neat materials from milligram to multi-gram; solutions from 1 mL ampoules/vials to custom volumes.
- Tamper-evident, inert packaging with lot-specific labels and barcodes for LIMS integration.
- Custom concentrations, blends, and isotope enrichments available upon request to match your method and instrument needs.
Instrument Compatibility
- Validated for LC-MS/MS, HPLC/UPLC (UV/FLD), GC-MS (after derivatization), and CE methods.
- Low-adsorption packaging and solvent systems selected to minimize carryover and adsorption on tubing and vials.
Support & Services
- Technical consultation for method setup, calibration design, and uncertainty budgets.
- Custom development of matrix-matched and multi-analyte reference materials for high-throughput laboratories.
- Stability and requalification services for extended studies and long-term projects.


