New user? / Forgot your password?
0 Item | 0,00 €
Pyraflufen (free acid) — Reference Materials for Residue Analysis
Pyraflufen free acid Precision Reference Materials for Confident Compliance
Drive defensible results in food and environmental residue testing with highpurity pyraflufen free acid reference materials from HPC Standards. Our ISO-aligned products deliver traceable calibration, robust method validation, and reliable LCMSMS confirmation at low gkg action limits. Available as neat materials or readytouse solutionswith optional isotopelabeled derivativesour portfolio supports matrix-matched calibration, spike-recovery studies, and proficiency testing. Benefit from comprehensive CoAs, batch-to-batch consistency, short lead times, and expert technical support to keep your laboratory audit-ready and compliant with global MRLs.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 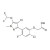 | 675714 | 1X10MG | Please log in. |
ISO 17034 Certified Reference Material | 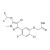 | 694318 | 1X5MG | Please log in. |
High-purity reference materials of pyraflufen (free acid) for robust quantification, method validation, and regulatory compliance in food and environmental residue testing.
Overview
Pyraflufen (free acid) is the de-esterified metabolite of the herbicide pyraflufen-ethyl, belonging to the protoporphyrinogen oxidase (PPO) inhibitor class. It is a key analyte in pesticide residue monitoring because it can occur as a metabolite in treated crops and environmental samples.
As a target compound in multi-residue methods, pyraflufen (free acid) requires reliable calibration and confirmation for compliance with international maximum residue limits (MRLs).
Uses
Pyraflufen-ethyl (parent ester) is applied post-emergence for broadleaf weed control and as a desiccant/defoliant in various crops. Hydrolysis of the ester leads to pyraflufen (free acid) as a major residue component relevant for enforcement.
Typical matrices include cereals, oilseeds, fruits, vegetables, and environmental compartments such as surface water and agricultural soils.
Mode of Action
Pyraflufen inhibits PPO (protoporphyrinogen oxidase), causing accumulation of photoreactive porphyrins, membrane lipid peroxidation, and rapid necrosis of plant tissue. This biochemical mode of action informs metabolite profiling and selection of analytical transitions in LC-MS/MS.
Regulatory Landscape
Residue definitions for enforcement may include pyraflufen (free acid) and, depending on jurisdiction, related metabolites. Global authorities (e.g., EU, Codex, US) publish crop-specific MRLs and guidance values.
Laboratories require validated methods capable of meeting action limits in the low µg/kg range, with appropriate identification points and confirmatory transitions.
Maximum Residue Limits (MRLs) and Compliance
MRLs vary by crop, region, and residue definition. Verify current limits with the relevant authority database prior to analysis and reporting.
Use traceable reference materials for calibration and quality control to ensure defensible data at or below regulatory decision thresholds.
Monitoring and Analytical Methods
LC-MS/MS is commonly used for pyraflufen (free acid) due to its polarity and ionization behavior; negative electrospray ionization (ESI−) is frequently selected with compound-specific MRM transitions. High-resolution MS may be used for confirmation.
Quantification typically employs matrix-matched calibration and, where available, isotopically labeled internal standards to correct for suppression and recovery.
Sample Preparation and Matrices
QuEChERS-based workflows (extraction with acetonitrile, buffered salts) followed by d-SPE cleanup are widely applied for food matrices. For soil and water, solid–liquid extraction and SPE concentration steps are common.
Matrix effects can be significant in high-pigment commodities; rigorous QC (blanks, spikes, duplicates) is recommended to maintain method performance.
Metabolites and Degradation Products
Pyraflufen (free acid) is a primary hydrolysis product of pyraflufen-ethyl and may co-occur with additional transformation products depending on environmental conditions (hydrolysis, photolysis, and microbial processes).
Inclusion of the free acid in residue definitions and analytical scope is critical for comprehensive compliance monitoring.
Human Toxicity
Available data for the active substance class indicate low to moderate acute mammalian toxicity with potential for eye/skin irritation. Risk characterization is exposure-dependent and regulated via MRLs and operator exposure limits.
Laboratory handling should follow chemical hygiene standards; avoid inhalation, ingestion, and skin contact.
Environmental Impact
PPO inhibitors can be hazardous to non-target plants and aquatic organisms. Pyraflufen-related residues may undergo photolysis and microbial degradation with matrix- and climate-dependent dissipation.
Environmental monitoring focuses on run-off, surface waters, and adjacent habitats to assess off-target exposure.
Safety Measures and Handling
Wear appropriate PPE (lab coat, nitrile gloves, eye protection). Handle solutions in a fume hood. Avoid sources of contamination when preparing calibrants and QC samples.
Dispose of waste via approved hazardous chemical routes consistent with local regulations.
Storage and Stability
Store neat materials and solutions protected from light at controlled temperature per certificate guidance. Use inert solvents of suitable purity. Prepare fresh working solutions at defined intervals to ensure stability.
Document lot numbers, preparation dates, and storage conditions to support data traceability.
Quality and Traceability
HPC Standards provides reference materials manufactured and tested to international quality requirements with comprehensive certificates of analysis (purity, identity, uncertainty, and traceability where applicable).
Batch-to-batch consistency supports long-term method performance and comparability across studies and sites.
Applications in Food and Environmental Laboratories
Suitable for calibration curves, system suitability, spike-recovery studies, matrix-matched standards, and proficiency testing preparations.
Supports confirmation at regulatory decision points and verification of method accuracy, precision, LOQ, and measurement uncertainty.
Analytical Considerations
Recommended to use isotopically labeled internal standards when available to correct for matrix effects and extraction variability.
Select ion transitions and collision energies validated for your instrument platform; confirm identity with retention time and ion ratio criteria.
Analytical Standards and Reference Materials
HPC Standards offers pyraflufen (free acid) reference materials and, on request, related metabolites and stable isotope-labeled derivatives for internal standardization.
Custom concentrations, solvent systems, and multi-analyte mixes can be provided to match your validated method.
Product Portfolio at HPC Standards
Available as neat material and as ready-to-use solutions for LC-MS/MS workflows. Each item includes a certificate of analysis and handling instructions.
Tailored reference materials for pesticide residues, veterinary drugs, metabolites, and isotope-labeled analogs enable end-to-end method development and routine monitoring.
Ordering and Support
Short lead times, temperature-controlled shipping, and global delivery. Technical support available for method setup, stability, and documentation requirements.
Contact our application specialists for assistance with method-specific mixes and accreditation-ready documentation.


