New user? / Forgot your password?
0 Item | 0,00 €
Bixafen desmethyl – Reference Materials for Residue Analysis
Bixafen desmethyl Precision Reference Materials for Confident Compliance
Drive reliable LCMSMS quantification with Bixafen desmethyl reference materials from HPC Standards. Our high-purity, traceable solutions and neat standards support sensitive residue analysis in food and environmental matricesaligned with international quality requirements. Ensure accurate calibration, confirmatory ion ratios, and matrix-matched performance at low gkg levels. Delivered with CoA, purity profile, and stability data, our materials empower method development, routine monitoring, and proficiency testingso your lab meets evolving regulatory limits with certainty.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Certified Reference Material Bixafen desmethyl solution | 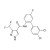 | 688024 | 1X1ML | Please log in. |
High-quality reference materials for Bixafen desmethyl to support accurate quantification in food and environmental residue analysis. Manufactured and tested to international quality requirements for reliable compliance monitoring.
Overview
Bixafen desmethyl is a primary metabolite of the succinate dehydrogenase inhibitor (SDHI) fungicide Bixafen. It is formed via N-demethylation and can occur in crops, soil, and water following agricultural use of Bixafen. Due to its relevance in residue definitions and risk assessments, Bixafen desmethyl is an important target analyte in regulatory monitoring.
As a structurally related transformation product, Bixafen desmethyl may share analytical characteristics with the parent compound, necessitating robust, matrix-validated methods and traceable reference materials for precise identification and quantification.
Uses (Analytical Context)
While Bixafen desmethyl is not an applied active substance, it is routinely monitored as:
- A target analyte in pesticide residue testing of plant-derived foods, processed commodities, and animal matrices where relevant.
- A marker for environmental fate and behavior studies (soil/water transformation, mobility, and persistence).
- A component in method validation and proficiency testing for LC–MS/MS multi-residue workflows.
Regulatory Landscape
Regulatory authorities may include Bixafen metabolites (such as desmethyl derivatives) in residue definitions for risk assessment and/or enforcement, depending on jurisdiction and commodity. Maximum residue limits (MRLs) and environmental quality standards are set at low µg/kg to mg/kg levels, requiring sensitive, selective analytical methods.
Monitoring requirements can encompass raw and processed foods, primary crops with SDHI applications, rotational crops, and environmental compartments. Laboratories should consult the latest regional regulations and guidance documents to confirm residue definitions, LOQs, and method performance criteria.
Monitoring and Surveillance
Routine monitoring programs typically implement LC–MS/MS screening and quantification for Bixafen and relevant metabolites including Bixafen desmethyl. Performance targets include:
- Method LOQs at or below applicable MRLs.
- Confirmatory ion ratios and retention time matching against certified reference materials.
- Ongoing quality control with matrix-matched calibration, internal standards where available, and participation in proficiency tests.
Health Impact
Risk assessments of Bixafen consider relevant metabolites such as Bixafen desmethyl. Toxicological evaluations focus on whether the metabolite shares the parent’s mode of action (SDHI) and its comparative potency. Human exposure may occur via dietary intake; hence conservative residue definitions and robust analytical verification are applied to protect consumers.
Human Toxicity
Available data for Bixafen desmethyl are typically derived from the parent compound’s dossier and metabolite-specific studies. Key considerations include systemic toxicity, potential for cumulative effects within the SDHI class, and margins of exposure based on dietary intake estimates. Laboratories must ensure analytical coverage of relevant metabolites to support accurate risk characterization.
Environmental Impact
Bixafen desmethyl can be detected in soil and water following degradation and transformation of Bixafen. Environmental fate assessments examine persistence, mobility (including potential for leaching), and degradability. Monitoring programs in surface waters and groundwater may include the metabolite where exposure is plausible.
Effects on Wildlife
Ecotoxicological relevance is evaluated in conjunction with the parent compound. Potential effects on non-target organisms (aquatic invertebrates, fish, soil organisms, and plants) are assessed based on predicted environmental concentrations and metabolite-specific hazard data where available.
Safety Measures (Laboratory Handling)
- Handle Bixafen desmethyl reference materials in a fume hood with appropriate PPE (lab coat, nitrile gloves, safety glasses).
- Avoid inhalation, ingestion, and skin contact; prevent environmental release.
- Follow site-specific chemical hygiene plans and waste disposal regulations.
- Refer to the product SDS for detailed hazard and precautionary statements.
Analytical Methods
- Primary techniques: LC–MS/MS or UHPLC–MS/MS with ESI, employing multiple reaction monitoring and matrix-matched calibration.
- Sample preparation: QuEChERS-based workflows or tailored SPE/cleanup depending on matrix complexity (high-fat, high-water, or acidic matrices).
- Identification/confirmation: Two transitions minimum, ion ratio tolerance per regulatory guidance, and retention time within validated windows.
- Validation: Linearity across low µg/kg to mg/kg ranges, recovery (70–120% typical), precision (RSD within guidance), and inter-day robustness.
Reference Materials
HPC Standards provides high-purity Bixafen desmethyl reference materials for quantitative residue analysis. Materials are produced and tested according to international quality requirements and meet the highest industrial standards. Batch-specific documentation includes certificate of analysis, purity profile, and stability information.
Available formats include neat solids and ready-to-use solutions in common LC–MS compatible solvents at defined concentrations with low uncertainty to support calibration, system suitability, and QC.
Quality and Compliance
Our reference materials are designed to support compliance with international regulations for pesticide residues in food and environmental matrices. Each lot undergoes rigorous identity confirmation and purity assessment using orthogonal techniques to ensure traceable, reproducible results in accredited laboratory workflows.
Storage and Stability
- Store sealed containers at recommended temperatures (typically refrigerated or as stated on the label) and protect from light and moisture.
- Allow solutions to equilibrate to room temperature before opening to minimize condensation.
- Observe expiry dates and re-test intervals specified on the certificate of analysis.
Applications
- Method development and validation for multi-residue LC–MS/MS.
- Routine quantification in official control and industry laboratories.
- Matrix fortification for recovery studies and measurement uncertainty estimation.
- Proficiency testing and internal quality control.
Matrices and Typical Levels
Applicable to a wide range of commodities: cereals, oilseeds, fruits, vegetables, processed foods, feeds, soils, surface and groundwater. Typical monitoring ranges: low µg/kg (sub-ppb) to low mg/kg, aligned with regulatory MRLs and environmental quality thresholds.
Documentation and Support
Each Bixafen desmethyl reference material is supplied with comprehensive documentation (CoA, safety data, handling and storage recommendations). Our technical team supports method optimization, matrix selection, and uncertainty considerations for demanding residue applications.
Regulatory Notes
Residue definitions and MRLs may evolve; laboratories should consult current regional legislation and guidance to confirm if Bixafen desmethyl is included for enforcement or risk assessment in specific commodities. HPC Standards provides consistent reference materials to maintain continuity across regulatory updates.


