New user? / Forgot your password?
0 Item | 0,00 €
Maltose Monohydrate Reference Materials
Maltose monohydrate Precision Reference Materials for Confident Quantification
Accelerate your carbohydrate analytics with high-purity maltose monohydrate reference materials from HPC Standards. Rigorously characterized and supplied with a comprehensive CoA, our materials ensure traceable calibration, robust method validation, and reliable routine QC across food, feed, pharmaceutical, and environmental labs. Available as crystalline solid or ready-to-use aqueous ampoules, each lot is tested to international quality requirements to support ISOIEC 17025 workflows. Benefit from consistent performance in HPLC-RID, HPAEC-PAD, enzymatic assays, and derivatized LC-MS applicationsplus custom concentrations, bulk supply, and multi-analyte kits on request. Choose HPC Standards for dependable results, minimized prep error, and compliance you can document.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
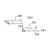 | 680275 | 1X250MG | Please log in. |
High-purity maltose monohydrate reference materials for calibration, method validation, and routine QC in food, feed, pharmaceutical, and environmental laboratories.
Overview
Maltose monohydrate is a reducing disaccharide (glucose–glucose, α(1→4)) containing one molecule of crystallization water. It is widely used as a quantitative marker in carbohydrate profiling and process monitoring. HPC Standards provides rigorously characterized reference materials to support traceable quantification and method performance.
Typical applications include HPLC-RID/HPAEC-PAD quantitation of sugars in foods and beverages, fermentation monitoring, excipient identification in pharmaceuticals, and research on carbohydrate metabolism.
Chemical Identity
CAS: 6363-53-7
Synonyms: 4-O-α-D-Glucopyranosyl-D-glucose monohydrate; Malt sugar monohydrate
Molecular formula: C12H24O12 (C12H22O11·H2O)
Molar mass: ~360.31 g/mol (monohydrate)
Key properties: highly water-soluble; reducing sugar; hygroscopic; prone to Maillard reactions with amines at elevated temperature.
Uses
- Calibration and system suitability for carbohydrate methods (food labeling sugars, brewing, dairy, confectionery).
- Process control in starch hydrolysis and fermentation (degree of saccharification, sugar balance).
- Excipient identification and assay in pharmaceutical formulations.
- Research standards for enzymology (amylase, maltase) and metabolic studies.
Regulatory Status
Maltose is widely used in foods and is generally recognized as safe (GRAS) in many jurisdictions when used as intended. For analytical use, laboratories should follow applicable regulations (e.g., ISO/IEC 17025) and internal validation requirements.
Reference materials from HPC Standards are manufactured and tested according to stringent international quality requirements and supplied with detailed documentation to support regulatory compliance.
Monitoring and Analytical Applications
- Food and beverage: quantification in cereals, syrups, beer, and baked goods.
- Clinical and nutrition research: carbohydrate profiling in model systems and formulations.
- Environmental and bioprocess: monitoring sugar release/consumption in biodegradation and fermentation matrices.
Health Impact
At analytical handling levels, maltose monohydrate exhibits low acute toxicity. As a dietary carbohydrate, excessive intake contributes to caloric load and may influence glycemic response.
Occupational exposure risks are minimal; dust may cause transient irritation in sensitive individuals.
Environmental Impact
Maltose is readily biodegradable and exhibits low environmental persistence. It does not bioaccumulate and has low aquatic toxicity at environmentally relevant concentrations.
Safety Measures
- Avoid dust generation; use local exhaust where appropriate.
- Wear standard laboratory PPE (lab coat, safety glasses, gloves).
- First aid: rinse with water after eye/skin contact; move to fresh air if dust inhaled.
Analytical Reference Materials
HPC Standards supplies maltose monohydrate reference materials with defined purity and comprehensive characterization. Each lot is accompanied by a Certificate of Analysis (CoA) detailing purity (e.g., HPLC/enzymatic), water content, identity confirmation (IR/NMR/MS as applicable), and uncertainty information to support traceable calibration.
Available formats include crystalline solid and ready-to-use aqueous ampoules at specified concentrations to minimize preparation error.
Sample Preparation
- Dissolve accurately weighed solid in ultrapure water; filter (0.22–0.45 µm) if required.
- For LC with high organic proportion, prepare in water and mix with mobile phase to prevent precipitation; sugars have limited solubility in high acetonitrile.
- Use gravimetric preparation and record temperature for density corrections when preparing calibrators.
Analytical Methods
- HPLC-RID: Amino, amide, or HILIC phases; isocratic water/acetonitrile; typical LOD low µg/mL.
- HPAEC-PAD: Strong anion-exchange with NaOH/acetate gradients for high selectivity among oligo/disaccharides.
- LC-MS/MS: Possible with derivatization (e.g., PMP, 2-AB) to enhance ionization and selectivity.
- Enzymatic assays: Specific hydrolysis followed by glucose quantification for confirmatory measurements.
Calibration and Traceability
- Prepare multi-point calibration (≥5 levels) bracketing expected sample concentrations.
- Verify linearity, precision, recovery, and matrix effects; include quality control samples at low/mid/high levels.
- Use internal standards where applicable (e.g., stable isotope-labelled glucose for hydrolysis control or matrix compensation).
Stability and Storage
- Store solid reference materials tightly closed, dry, and protected from light at 2–8 °C; minimize headspace humidity (desiccant recommended).
- Aqueous solutions are stable refrigerated for limited periods; follow CoA for shelf life. Avoid microbial contamination; use sterile filtration for long runs.
- Minimize freeze–thaw cycles; aliquot as needed.
Matrix Effects and Interferences
- Reducing sugars may undergo degradation or Maillard reactions in complex matrices; control temperature and pH.
- Coelution with other saccharides can occur; use selective columns or PAD detection to resolve isomers.
- High salt or protein content may affect detector response; consider sample cleanup (dilute-and-shoot, SPE desalting, or precipitation).
Packaging and Formats
- Solid: preweighed vials for gravimetric solution preparation.
- Solution: ready-to-use aqueous ampoules at certified concentrations to reduce preparation uncertainty.
Custom concentrations, bulk quantities, and multi-analyte carbohydrate calibration kits are available on request.
Compliance and Documentation
Each reference material is supplied with a CoA, safety data sheet (SDS), recommended storage/handling instructions, and detailed method notes to support ISO/IEC 17025 workflows and internal SOPs.
Related Products
- Other carbohydrates: glucose, fructose, sucrose, lactose, maltotriose.
- Derivatization reagents and eluents for HPAEC-PAD/HILIC methods.
- Stable isotope-labelled sugars on request for advanced quantification strategies.
FAQs for Laboratories
Q: Do I need derivatization? A: Not for RID/HPAEC-PAD; derivatization may be useful for LC-MS sensitivity.
Q: How to avoid microbial growth in standards? A: Prepare fresh, use sterile filtration, refrigerate, and adhere to CoA stability limits.
Q: Can I use anhydrous maltose instead? A: Yes, but account for molecular weight and water-of-crystallization differences when calculating concentrations.
Ordering Information
- Product: Maltose monohydrate reference material
- CAS: 6363-53-7 | Matrix: solid or aqueous | Purity: specified on CoA
Contact HPC Standards for catalog numbers, pack sizes, custom concentrations, and lead times.


