New user? / Forgot your password?
0 Item | 0,00 €
Florfenicol Reference Materials
Florfenicol Precision Reference Materials for Confident Compliance
Elevate your LCMSMS workflows with certified, traceable Florfenicol reference materials from HPC Standards. Our high-purity neat and ready-to-use solutionsincluding florfenicol amine and isotope-labelled optionsdeliver robust calibration, spike-recovery, and QC across food, feed, and environmental matrices. Produced under international quality systems and supplied with comprehensive CoAs, our materials help accredited labs achieve low-ppb LOQs, meet stringent MRLs, and safeguard data integrity. Custom concentrations and multi-residue mixes availableoptimize your method, ensure reliable results, and accelerate regulatory readiness.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 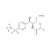 | 694359 | 1X5MG | |
ISO 17034 Certified Reference Material D3-Florfenicol solution | 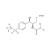 | 694360 | 1X1ML | |
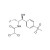 | 674759 | 1X250MG | Please log in. |
High-quality Florfenicol reference materials for robust residue analysis in food, feed, and environmental matrices. Certified, traceable, and optimized for LC–MS/MS workflows to ensure compliance and data integrity.
Overview
Florfenicol is a synthetic broad-spectrum amphenicol-class antimicrobial used exclusively in veterinary medicine. It is structurally related to chloramphenicol and thiamphenicol, but designed to reduce adverse effects and resist common bacterial inactivation mechanisms. It is widely applied in terrestrial livestock and aquaculture for treatment and metaphylaxis of bacterial infections.
In residue analysis, the marker residue often includes the parent compound and the primary metabolite florfenicol amine (FFA). Laboratories routinely monitor florfenicol to verify compliance with Maximum Residue Limits (MRLs) and withdrawal periods.
Uses in Veterinary Medicine
Indications include respiratory and systemic infections in cattle, swine, and poultry, and bacterial diseases in farmed fish and shrimp. Florfenicol is not approved for human use. Routes of administration include feed medication, in-water dosing for aquaculture, and parenteral formulations.
Its veterinary-only status requires targeted residue surveillance in animal-derived food products and aquatic products to protect consumers and support export compliance.
Regulatory Status and MRLs
MRLs/tolerances for florfenicol are established in many jurisdictions (e.g., EU, Codex, US) for matrices such as muscle, liver, kidney, fat/skin, milk, and aquaculture products. Marker residue definitions commonly comprise florfenicol and/or the sum of florfenicol and florfenicol amine. Laboratories should consult the latest regulations (e.g., EU Reg. 37/2010 and subsequent amendments, Codex MRLs, FDA tolerances) for matrix-specific limits and withdrawal times.
Import controls and third-country requirements may mandate specific analyte panels and reporting limits; method validation must reflect the most stringent applicable regulations.
Monitoring and Compliance
National residue control plans and industry QA programs include routine monitoring of florfenicol in raw and processed products. Typical surveillance matrices include bovine/porcine/poultry tissues, milk, eggs, honey (where relevant), as well as fish and crustaceans.
Accredited laboratories employ validated methods with defined decision limits (CCα) and detection capabilities (CCβ), supported by fit-for-purpose reference materials for calibration, spiking, and quality control.
Mechanism of Action and Resistance
Florfenicol inhibits bacterial protein synthesis by binding to the 50S ribosomal subunit (peptidyl transferase center). It is active against many Gram-negative and Gram-positive pathogens relevant to veterinary and aquaculture settings.
Resistance can arise via efflux pumps, ribosomal protection/mutation, and acquisition of amphenicol resistance genes. Prudent use and effective residue monitoring help mitigate selection pressure and dissemination of resistance determinants.
Human Toxicity and Occupational Exposure
Florfenicol is not intended for human therapy. Toxicological evaluations inform regulatory MRLs and Acceptable Daily Intakes (ADI). Compared with chloramphenicol, florfenicol has a reduced risk profile, yet laboratory handling should minimize exposure via dermal contact, inhalation of aerosols, or ingestion.
Use appropriate PPE, implement good laboratory hygiene, and follow safety data sheet (SDS) guidance when preparing analytical solutions and handling reference materials.
Environmental Impact
Residues can enter aquatic and terrestrial environments via medicated feed, excreta, and aquaculture effluents. Florfenicol and its metabolites may persist to varying degrees, with potential effects on microbial communities and selection for antimicrobial resistance.
Environmental monitoring focuses on water, sediment, and biota in aquaculture regions and downstream areas, using sensitive LC–MS/MS methods and matrix-matched calibration.
Effects on Wildlife and Microbiota
Non-target effects in aquatic organisms can include sublethal impacts on growth, behavior, and microbiome composition. Changes to sediment and water microbial communities may alter ecosystem functions and resistance gene prevalence.
Risk assessments incorporate exposure modeling, degradation kinetics, and species-specific toxicity data to guide stewardship and regulatory measures.
Physicochemical Properties
Class: Amphenicol antibiotic; typical features include moderate lipophilicity and limited volatility. Solubility and stability are solvent- and pH-dependent, relevant for extraction and chromatographic performance.
Reference solutions are commonly prepared in acetonitrile, methanol, or mixed aqueous-organic media. Final mobile phases should ensure adequate retention, peak shape, and ionization efficiency in MS.
Analytical Methods
Primary technique: LC–MS/MS in MRM mode for parent and metabolites (e.g., florfenicol and florfenicol amine). HPLC-UV may be used for select matrices but offers lower selectivity/sensitivity.
Sample preparation: protein precipitation or QuEChERS-style extraction, often followed by SPE cleanup (e.g., polymeric or mixed-mode sorbents). Matrix-matched calibration and isotopically labeled internal standards improve quantitation and compensate for ion suppression/enhancement.
Sample Matrices and Typical Performance
Matrices: muscle, liver, kidney, fat/skin, milk, eggs, fish, shrimp, feed, water, and sediment. Methods typically target low µg/kg (ppb) levels in food matrices and ng/L–µg/L in environmental samples, subject to regulatory or program-specific LOQs.
Method validation should include selectivity, linearity, recovery, precision, measurement uncertainty, stability (bench/auto-sampler), and robustness across matrix lots.
Reference Materials from HPC Standards
HPC Standards GmbH supplies certified reference materials for florfenicol and key related analytes to support calibration, spike-recovery, and quality control in residue testing. Products are manufactured under stringent quality requirements and accompanied by detailed Certificates of Analysis (CoA).
Available formats include neat materials and ready-to-use solutions, as well as stable isotope-labeled reference materials (where applicable) for internal standardization in LC–MS/MS methods.
Related Compounds and Metabolites
Common analytes in regulatory scope: florfenicol (parent), florfenicol amine (FFA, primary metabolite), and occasionally other amphenicols for extended screening (e.g., chloramphenicol, thiamphenicol) as prohibited or controlled comparators depending on jurisdiction.
In some regulatory frameworks, reporting may require the sum of parent plus metabolite; laboratories should align target lists and calibration strategies accordingly.
Quality Assurance, Certification, and Traceability
HPC reference materials are produced and released under internationally recognized quality systems. CoAs document identity, purity or assigned value, expanded uncertainty, stability information, and metrological traceability (where applicable to SI units).
Batches are homogeneity- and stability-verified to ensure reliable performance across their stated shelf life.
Storage, Handling, and Safety Measures
Store reference materials according to the CoA (typically refrigerated or frozen; protect from light and moisture). Allow solutions to equilibrate to room temperature before opening to avoid condensation. Use clean, inert labware and minimize freeze–thaw cycles.
Follow SDS recommendations: use gloves, lab coat, eye protection; avoid inhalation and ingestion; dispose of waste in accordance with institutional and legal requirements.
Documentation, Shipping, and Support
Each shipment includes a CoA and SDS. Temperature-controlled logistics are available for temperature-sensitive products. Technical support provides method guidance for LC–MS/MS setup, internal standard selection, and matrix-specific considerations.
Custom concentrations, mixtures (multi-residue panels), and isotopically labeled reference materials can be provided upon request to match your method and regulatory needs.
Applications and Compliance
Typical applications: routine MRL compliance testing in accredited laboratories, import/export verification, industry QA programs, proficiency testing, and environmental surveillance in aquaculture regions.
HPC reference materials enable defensible quantitation, facilitate accreditation (e.g., ISO/IEC 17025), and support consistent decision-making at regulatory thresholds.


