New user? / Forgot your password?
0 Item | 0,00 €
Decoquinate
Decoquinate reference materials Precision you can trust. Elevate your residue analysis with HPC Standards high-purity, certified Decoquinate CRMs for LC-MSMS and HPLC workflows. Our neat materials and ready-to-use solutions support calibration, method validation, and routine QC across tissues, milk, eggs, feed, and environmental matrices. Each lot ships with a comprehensive CoA traceability, uncertainty to meet international quality requirements and help your lab demonstrate compliance with MRLs. Request custom concentrations, multi-analyte mixes, and fast deliveryoptimized for robust recoveries, low LOQs, and confident regulatory reporting.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 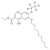 | 679567 | 1X10MG | Please log in. |
D5-Decoquinate solution | 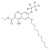 | 690896 | 1X1ML | Please log in. |
ISO 17034 Reference Material | 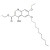 | 674876 | 1X100MG | Please log in. |
High-quality reference materials for precise determination of decoquinate residues in food and environmental matrices. Ensure regulatory compliance and confident method validation with HPC Standards.
Overview
Decoquinate is a quinoline-class coccidiostat used in veterinary medicine and as a feed additive to prevent coccidiosis in poultry and ruminants. It inhibits mitochondrial electron transport in protozoa, leading to parasite death. Low water solubility and high lipophilicity influence its distribution and residue behavior.
In residue analysis, decoquinate is a target analyte in tissues (e.g., liver, muscle), milk, eggs (where applicable), and feed. Reliable quantification supports food safety and compliance with established maximum residue limits (MRLs).
Uses and Mode of Action
Primary use: prophylaxis of coccidiosis caused by Eimeria spp. in chickens, turkeys, and ruminants. Often administered via medicated feed.
Mode of action: inhibition of the mitochondrial cytochrome bc1 complex (electron transport), disrupting ATP synthesis in susceptible protozoa.
Chemistry and Physicochemical Properties
Chemical class: quinoline carboxylate ester coccidiostat. CAS: 18507-89-6; Formula: C24H35NO5.
Key traits: very low aqueous solubility, high log Kow, strong adsorption to organic matter; thermally stable under typical analytical conditions. These properties favor LC-based methods with organic extraction and cleanup.
Regulatory Status and MRLs
Decoquinate is regulated as a veterinary medicinal product/feed additive in many jurisdictions. MRLs for edible tissues (e.g., liver, kidney, muscle, fat) and, where authorized, milk/eggs, are established by authorities (e.g., EU, US, other national agencies).
Compliance requires laboratories to apply validated methods meeting regulatory performance criteria (specificity, accuracy, precision, recovery, LOQ).
Monitoring and Compliance
Targeted monitoring covers medicated feeds, carry-over in non-target feeds, and residues in foods of animal origin. Risk-based surveillance focuses on high-exposure matrices (liver) and withdrawal verification.
Routine QA includes use of matrix-matched calibration, independent reference materials, internal standards, and participation in proficiency testing.
Analytical Methods
Preferred techniques: LC-MS/MS for confirmatory analysis; HPLC-UV/FLD for screening when sensitivity and selectivity allow. Sample prep often employs QuEChERS-type extraction or SPE cleanup due to lipophilicity.
Typical performance: sub-µg/kg to low µg/kg LOQs in tissues and milk with MS/MS; broader linear ranges and robust recoveries validated per SANTE/ISO/ICH guidance.
Reference Materials from HPC Standards
HPC Standards supplies certified reference materials for decoquinate to support calibration, system suitability, and quality control across diverse matrices.
Available formats: neat materials and ready-to-use solutions at custom concentrations; options for multi-analyte mixes on request. Each lot includes detailed documentation (CoA, uncertainty, traceability).
Matrices and Typical Residue Levels
Common matrices: poultry and ruminant liver, muscle, fat, kidney; milk (if authorized use); eggs (jurisdiction-dependent); feed and premixes; environmental samples near livestock operations.
Residue occurrence is influenced by dose, species, metabolism, and withdrawal time; liver often exhibits higher concentrations than muscle. Laboratories should align LOQs with applicable MRLs.
Human Toxicity and Risk Assessment
Decoquinate exhibits low acute toxicity and low systemic bioavailability; available evaluations have not indicated genotoxic or carcinogenic concerns at permitted exposures. Risk management relies on ADI-based MRLs and adherence to withdrawal periods.
Laboratory handling risks are primarily related to dust exposure and solvent use; implement standard chemical hygiene and PPE.
Environmental Impact and Ecotoxicology
Due to high hydrophobicity, decoquinate tends to adsorb to soils and sediments. Degradation rates depend on environmental conditions; mobility in groundwater is expected to be low, while aquatic exposure may affect sediment-dwelling organisms.
Risk assessments consider potential effects on aquatic invertebrates and algae; proper manure and effluent management reduces environmental load.
Effects on Wildlife
Indirect exposure may occur via contaminated feed, litter, or runoff. Monitoring near intensive livestock operations helps assess local ecological risk, with attention to benthic habitats and invertebrates.
Safety Measures and Handling
Handle reference materials with gloves, lab coat, and eye protection in a well-ventilated area or fume hood. Avoid generation of dust and inhalation. Use appropriate solvent-resistant equipment for solutions.
Dispose of waste according to local regulations for pharmaceutical/veterinary compounds and organic solvents.
Storage and Stability
Store sealed containers in a cool, dry, and dark place. Avoid prolonged exposure to light and elevated temperatures. Follow the product’s shelf-life and re-test intervals as stated on the Certificate of Analysis.
Solution standards should be stored at recommended temperatures (typically 2–8°C) and used within the stated stability period.
Quality and Accreditation
HPC Standards produces reference materials under stringent quality systems aligned with international requirements to ensure traceability, homogeneity, and defined measurement uncertainty.
Batch-specific CoAs accompany every product, supporting accreditation needs (e.g., ISO/IEC 17025) and regulatory submissions.
Related Compounds and Metabolites
Analytical scopes may include structurally related coccidiostats and relevant decoquinate metabolites depending on jurisdictional requirements and metabolic pathways.
Contact us for custom mixes covering multi-residue panels for feed additives and veterinary drugs.
Applications in Food and Environmental Labs
Use cases: method development and validation, routine QC in residue testing, proficiency testing support, instrument calibration, and troubleshooting.
Applicable sectors: contract testing laboratories, official control labs, feed manufacturers, veterinary pharmaceutical QC, and environmental monitoring units.
Documentation and Certificates
Each reference material is supplied with a Certificate of Analysis detailing identity, purity or assigned value, uncertainty, traceability, storage conditions, and expiry.
Supplemental documents (SDS, method notes) are available on request to streamline accreditation and audits.
Ordering Information
Product forms: neat materials and solution reference materials at tailored concentrations. Custom packaging and multi-analyte blends available.
For quotations, bulk orders, or technical support, contact HPC Standards. We provide fast lead times, technical guidance for method setup, and consistent lot-to-lot performance.


