New user? / Forgot your password?
0 Item | 0,00 €
Formylaminoantipyrine (FAA)
Formylaminoantipyrine Certified Reference Materials for Defensible Residue Results
Boost confidence in quantification with highpurity Formylaminoantipyrine FAA reference materials from HPC Standards. Purpose-built for ISOIEC 17025 workflows, our traceable FAA solutions and neat materials deliver precise calibration and QC across food, biological, and environmental matrices. Each batch is gravimetrically prepared, identity-verified by orthogonal techniques, and supplied with a comprehensive CoA to meet SANTE, VICH, and EMAFDA guidance. Choose HPC Standards for robust method performance, audit-ready documentation, and expert supportincluding custom concentrations, solvent systems, and multi-analyte mixes for metamizole metabolite panels.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 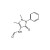 | 677051 | 1X10MG | Please log in. |
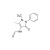 | 679632 | 1X10MG | Please log in. | |
D3-4-Formylaminoantipyrine solution | 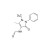 | 690906 | 1X1ML |
Precision reference materials for confident quantification of Formylaminoantipyrine (FAA), a major metabolite of metamizole (dipyrone), enabling robust residue analysis in food, feed, biological, and environmental matrices.
Overview
Formylaminoantipyrine (FAA) is a primary Phase I metabolite of the analgesic/antipyretic drug metamizole (dipyrone). FAA is routinely monitored as part of veterinary drug residue investigations and in environmental monitoring where pharmaceutical transformation products are of interest. Due to its polarity and occurrence as a transformation product, FAA requires sensitive and selective analytical workflows supported by high-quality reference materials.
HPC Standards GmbH provides traceable FAA reference materials specifically designed for quantitative residue analysis under ISO/IEC 17025 and related quality systems.
Uses
• Target analyte in veterinary drug residue testing related to metamizole usage in food-producing animals and horses (where applicable under jurisdictional rules).
• Metabolite marker in pharmacokinetic, toxicokinetic, and metabolism studies of metamizole.
• Indicator compound in environmental and wastewater-based monitoring of pharmaceutical transformation products.
Regulatory Context
• FAA is considered a relevant metabolite of metamizole and is often included in regulatory and surveillance methods for veterinary drug residues.
• Maximum residue limits (MRLs) and authorizations for metamizole vary by region and species; competent authorities may specify marker residues and target matrices (e.g., muscle, liver, kidney, milk, urine).
• Laboratories should verify current regional legislation, action limits, and residue definitions before reporting results.
Monitoring and Target Matrices
• Food and feed: edible tissues, milk, and offal from treated animals (as permitted regionally).
• Biological: plasma/serum, urine for PK/TK and residue depletion studies.
• Environmental: influent/effluent wastewater, surface water, sludge, and sediments in studies addressing pharmaceutical fate and occurrence.
Health Impact
Human toxicity (context via metamizole): Metamizole has known adverse effect concerns (e.g., rare agranulocytosis) in humans; FAA forms as a systemic metabolite. Toxicological data specific to FAA are more limited compared to parent/metabolites such as 4-aminoantipyrine. Risk assessments typically consider the full metabolite profile; FAA may be included in marker residue evaluations depending on jurisdiction.
Environmental Impact
• FAA can occur as a transformation product in aquatic systems where metamizole-related compounds are present. Reported occurrence depends on usage patterns and wastewater treatment efficiency.
• Environmental fate: polarity suggests mobility in water; biodegradation and photolysis may contribute to attenuation, but persistence can be matrix- and condition-dependent.
• Effects on wildlife: specific ecotoxicological data for FAA are limited; conservative monitoring is recommended where pharmaceutical residues are under scrutiny.
Safety Measures and Handling
• Handle FAA reference materials using standard laboratory PPE (lab coat, gloves, safety glasses) in a well-ventilated area or fume hood.
• Avoid skin and eye contact; prevent inhalation of aerosols/solvents; follow institutional chemical hygiene plans.
• Dispose of waste according to local regulations for pharmaceutical and organic solvent residues.
Analytical Methods
• Primary techniques: LC–MS/MS (MRM) for selectivity and sensitivity in complex matrices; HRMS for confirmation and suspect screening.
• Alternative/orthogonal: HPLC-UV/FLD possible in clean matrices or for semi-quantitative workflows, typically with derivatization or wavelength optimization.
• Sample preparation: protein precipitation (biofluids), SPE (reversed-phase or mixed-mode) for cleanup and enrichment; QuEChERS-based approaches adapted for veterinary drug residues in tissues and milk.
• Chromatography: reversed-phase columns (C18/C8), aqueous-compatible phases; gradient elution with acidified aqueous/organic mobile phases to improve peak shape and ionization.
• Validation: evaluate linearity, accuracy, precision, LOD/LOQ, recovery, matrix effects, selectivity, carryover, and stability according to SANTE, VICH, FDA/EMA, or ISO/IEC 17025 requirements.
Reference Materials
• HPC Standards FAA reference materials are produced for quantitative calibration and QC in accredited laboratories.
• Formats: neat material (where applicable) and ready-to-use certified solutions at defined mass fractions/concentrations to minimize preparation uncertainty.
• Traceability: gravimetrically prepared using calibrated balances; purity and identity verified by orthogonal techniques.
• Supplied with certificate of analysis (CoA) including purity, uncertainty, solvent system, concentration, and recommended storage conditions.
Quality and Compliance
• Manufactured under rigorous quality control aligned with international requirements for reference materials used in residue analysis.
• Suitable for use in methods compliant with ISO/IEC 17025 and relevant guidance documents (e.g., SANTE, VICH, EMA/FDA) where FAA is included as a target analyte.
• Batch-specific documentation supports audit and accreditation needs.
Storage and Stability
• Recommended storage: refrigerated or frozen conditions as indicated on the CoA (commonly 2–8 °C or ≤ –20 °C), protected from light in amber vials.
• Allow to reach room temperature before opening to avoid condensation; minimize headspace and repeated freeze–thaw cycles.
• Observe stated shelf life; verify stability in working solutions during method validation (short-term, long-term, autosampler).
Method Development Tips
• Optimize source parameters for positive ESI; monitor characteristic product ions for FAA to enhance selectivity in MRM.
• Use isotope-dilution where available to correct for matrix effects and extraction variability.
• Evaluate matrix-matched calibration or standard addition in complex tissues and milk.
• Employ system suitability checks with FAA reference materials prior to batch analysis.
Cross-Analyte Considerations
• FAA is commonly evaluated alongside related metamizole metabolites (e.g., 4-methylaminoantipyrine, 4-aminoantipyrine, 4-acetylaminoantipyrine) to meet residue definition requirements.
• Include appropriate internal standards and surrogates for the metabolite panel for robust quantification across matrices.
Matrices and Application Fields
• Food safety: surveillance of residues in meat, offal, milk, and gelatin where permitted by law.
• Veterinary studies: residue depletion and withdrawal time assessments.
• Environmental monitoring: occurrence and fate assessments in WWTP effluents and receiving waters.
Uncertainty and Traceability
• Concentration and purity assignments include expanded uncertainty with coverage factor k and confidence level specified on the CoA.
• Gravimetric preparation and purity assessment ensure metrological traceability to SI units where applicable.
Regulatory Reporting
• Confirm current regional residue definitions, MRLs, and action limits for metamizole-related analytes prior to reporting.
• Document method performance characteristics (LOQ, recovery, MU) for each matrix as part of the analytical report.
Product Specifications (typical)
• Available as neat or solution reference materials at defined concentrations in high-purity solvents (e.g., acetonitrile, methanol, or water mixtures).
• Identity confirmation by MS and NMR where applicable; purity assessed by chromatographic and spectroscopic methods.
• Packaged in amber glass vials with tamper-evident seals.
Ordering and Support
• Custom concentrations, solvent systems, and multi-analyte mixes (including metamizole metabolite panels) available on request.
• Technical dossiers, SDS, and CoA provided with shipment; method development support available from our scientific team.
• Global shipping with temperature-controlled logistics upon request to maintain material integrity.
Why HPC Standards
• Specialist manufacturer of reference materials for pesticides, veterinary drugs, metabolites, and stable isotope-labelled derivatives.
• Products tested according to international quality requirements and designed for the highest industrial standards.
• Reliable partner for food and environmental analysis ensuring regulatory compliance and defensible data.


