New user? / Forgot your password?
0 Item | 0,00 €
AOZ (3-amino-2-oxazolidinone) — Reference Materials for Nitrofurans Residue Analysis
AOZ Precision Reference Materials for Nitrofurans Compliance
Drive confident LC-MSMS confirmation at sub-ppb levels with AOZ 3-amino-2-oxazolidinone reference materials from HPC Standards. Our high-purity neat and solution standardsplus stable isotopelabelled AOZsupport method development, calibration, and routine QC across challenging matrices aquaculture, meat, dairy, eggs, honey. Manufactured and tested to international quality requirements, they enable laboratories to meet EU 2021808 and SANTE guidance, verify LOQCCCC, and deliver defensible results. Choose customizable concentrations, batchspecific documentation CoA, SDS, and proven stability for robust, compliant residue monitoring.
Product | Catalog No./ CAS No. | Quantity | Price | |
|---|---|---|---|---|
ISO 17034 Reference Material | 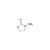 | 676836 | 1X50MG | Please log in. |
ISO 17034 Reference Material | 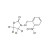 | 679160 | 1X10MG | Please log in. |
ISO 17034 Reference Material | 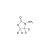 | 677256 | 1X10MG | Please log in. |
D4-AOZ solution |  | 690671 | 1X1ML | Please log in. |
High-quality AOZ reference materials for sensitive, compliant residue monitoring across food and environmental matrices. Manufactured and tested to international quality requirements to support robust LC-MS/MS confirmation and routine QC.
Overview
AOZ (3-amino-2-oxazolidinone) is the primary marker metabolite of the nitrofuran antibiotic furazolidone. In food-producing animals, nitrofurans form stable, protein-bound residues; AOZ is released by hydrolysis and quantified as an evidence of illegal administration. AOZ analysis is a cornerstone of veterinary drug residue surveillance in aquaculture, meat, dairy, eggs, and bee products.
Uses
- Target analyte for regulatory monitoring of nitrofuran residues (furazolidone exposure) in food of animal origin and related environmental samples.
- Method development, system suitability, calibration, and quality control in LC-MS/MS workflows (derivatization-based) using matched reference materials and stable isotope-labelled internal standards.
- Proficiency testing support and routine laboratory verification of LOQ/CCα/CCβ performance.
Regulatory
- European Union: Nitrofurans are prohibited in food-producing animals; marker metabolites (including AOZ) are controlled at very low action levels (typical Reference Points for Action around 0.5 µg/kg; verify current competent authority limits).
- Many jurisdictions apply zero-tolerance policies with action/decision limits commonly between 0.5–1.0 µg/kg for AOZ in edible tissues and aquaculture products.
- Method validation and confirmatory analysis should follow Commission Implementing Regulation (EU) 2021/808 and applicable SANTE/validation guidance for residues.
Monitoring
- Typical matrices: shrimp, fish, eels, soft-shell turtle, poultry, pork, beef, milk, eggs, honey, gelatin/collagen, animal feeds.
- Sampling plans prioritize aquaculture imports, high-risk supply chains, and processed protein matrices where protein-bound residues are expected.
- Routine objectives: achieve LOQs ≤0.5 µg/kg with confirmatory identification points and ion ratio tolerances per regulatory guidance.
Analytical Methods
- Principle: Release protein-bound AOZ by acid hydrolysis, derivatize with 2-nitrobenzaldehyde (2-NBA) to form the nitrophenyl derivative, and quantify by LC-MS/MS (MRM).
- Sample preparation: homogenization; acid hydrolysis; derivatization; liquid–liquid extraction and/or SPE cleanup (e.g., HLB/C18); matrix-matched calibration and isotope dilution.
- Instrumentation: Reversed-phase LC; ESI+ or ESI− depending on derivative; two MRM transitions for confirmation; monitor derivative stability and carryover.
Performance Characteristics
- Typical LOQ: 0.1–0.5 µg/kg (matrix-dependent). Calibration ranges commonly 0.05–5 µg/kg (as AOZ).
- Precision: intra-lab RSD generally ≤15% at action levels with isotope dilution.
- Uncertainty: evaluate contributions from derivatization efficiency, recovery, matrix effects, and reference material purity/concentration.
Human Toxicity
- AOZ is a marker of exposure to furazolidone. Nitrofurans and their metabolites have been associated with genotoxic and carcinogenic potential in experimental systems, underpinning global prohibitions in food-producing animals.
- Risk management is based on non-threshold (as low as reasonably achievable) control with stringent action limits.
Environmental Impact
- Parent nitrofurans are generally unstable in the environment; AOZ is primarily relevant as a metabolite within exposed animals and products. Environmental occurrence is low and data remain limited; food-chain monitoring is the principal control measure.
Safety Measures (Laboratory Handling)
- Handle AOZ reference materials with appropriate PPE (lab coat, gloves, eye protection) in a fume hood. Avoid inhalation and skin/eye contact.
- Derivatization reagents (e.g., 2-NBA) and acids are hazardous; follow chemical hygiene and waste disposal regulations.
- Prepare solutions gravimetrically using Class A glassware; document traceability and lot numbers for audits.
Method Validation & QA/QC
- Validate selectivity, linearity, recovery, matrix effects, precision, LOQ, CCα/CCβ, and stability per EU 2021/808 and relevant SANTE guidance.
- Include blanks, fortified matrix QC levels at/around the action limit, and independent check standards. Use stable isotope-labelled AOZ as internal standard when available.
Matrices & Interferences
- Complex protein matrices (e.g., gelatin/collagen) may influence hydrolysis and derivatization yields; optimize conditions and use matrix-matched calibration.
- Monitor potential artefacts and cross-reactivity from reactive carbonyls; verify identity with qualifier/quantifier ion ratios and retention time matching.
Storage & Stability
- Store AOZ neat materials protected from light and moisture; recommended 2–8 °C or −20 °C depending on formulation. Avoid repeated freeze–thaw of solutions.
- Use amber vials; assess solution stability (short- and long-term) and assign in-use shelf life per internal SOPs.
Documentation
- Each lot supplied with Certificate of Analysis (purity or concentration, traceability, uncertainty, intended use, storage, and expiry), safety data, and method guidance notes where applicable.
- Batch-specific characterization by appropriate techniques (e.g., LC, NMR, MS) under rigorous quality control aligned with international requirements.
Analytical Standards
- Use AOZ reference materials for calibration curves, system suitability, and ongoing QC to ensure regulatory compliance and defensible results.
- Stable isotope-labelled AOZ internal standards are recommended for isotope-dilution quantification and improved accuracy at sub-ppb levels.
HPC Standards Reference Materials
- AOZ neat and solution reference materials available with custom concentrations and package sizes for LC-MS/MS methods.
- Stable isotope-labelled AOZ reference materials for internal standardization and method validation.
- Products are tested according to international quality requirements and meet the highest industrial standards, supporting reliable, reproducible residue analysis in accredited laboratories.


